Fecal Microbiota Transplantation Market Size, By Region (North America, Europe, Asia-Pacific, Latin America, Middle East and Africa), By Statistics, Trends, Outlook and Forecast 2026 to 2033 (Financial Impact Analysis)
ID : MRU_ 444332 | Date : Feb, 2026 | Pages : 249 | Region : Global | Publisher : MRU
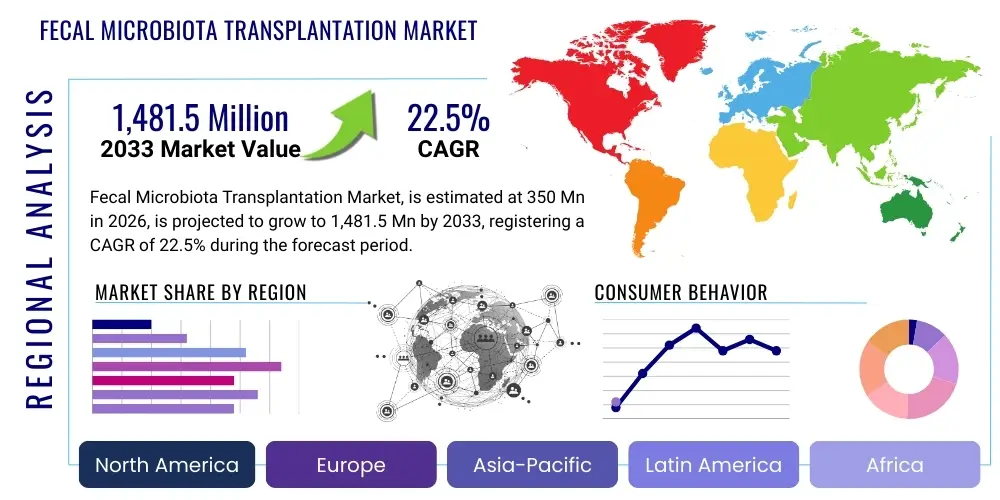
Fecal Microbiota Transplantation Market Size
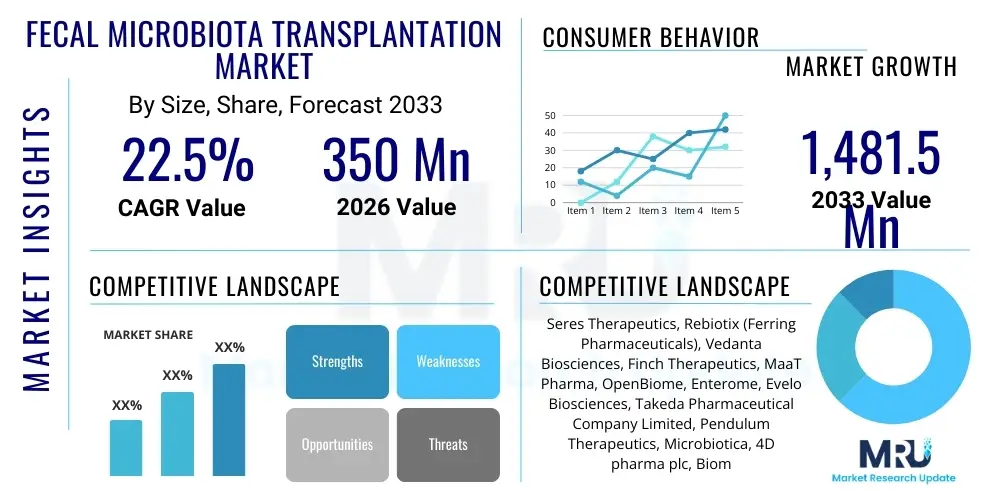
The Fecal Microbiota Transplantation Market is projected to grow at a Compound Annual Growth Rate (CAGR) of 22.5% between 2026 and 2033. The market is estimated at $350 Million in 2026 and is projected to reach $1,481.5 Million by the end of the forecast period in 2033.
Fecal Microbiota Transplantation Market introduction
The Fecal Microbiota Transplantation (FMT) market encompasses the development, production, and therapeutic application of fecal matter from a healthy donor into a recipient to restore a disrupted gut microbiome. This innovative medical procedure is primarily employed for the treatment of recurrent Clostridioides difficile infection (CDI), a severe and often life-threatening condition characterized by antibiotic-resistant bacterial overgrowth in the colon. Beyond CDI, significant research and clinical trials are actively exploring FMT's potential in addressing a myriad of gastrointestinal, metabolic, and autoimmune disorders, including inflammatory bowel disease (IBD), irritable bowel syndrome (IBS), autism spectrum disorders, and metabolic syndrome, thereby expanding its therapeutic landscape.
FMT involves the transfer of stool, containing a diverse community of microorganisms, from a carefully screened donor to a patient. This process aims to re-establish a healthy microbial balance within the recipient's gut, which is crucial for nutrient absorption, immune system modulation, and pathogen resistance. The primary benefits of FMT include its remarkable efficacy in treating recurrent CDI, often achieving cure rates exceeding 90%, and its potential to offer a novel therapeutic approach where conventional treatments have failed. The market is driven by several critical factors, including the increasing incidence and prevalence of recurrent CDI globally, the rising burden of antibiotic resistance, growing awareness among healthcare professionals and patients about microbiome-related health conditions, and substantial advancements in understanding the human gut microbiome's role in health and disease. Furthermore, ongoing clinical research, supportive regulatory pathways emerging in various regions, and the development of standardized administration methods and formulations are propelling market expansion. The versatility of FMT, coupled with its natural approach to restoring microbial equilibrium, positions it as a transformative modality in modern medicine, promising significant relief for patients suffering from persistent and debilitating gut dysbiosis-related conditions.
Fecal Microbiota Transplantation Market Executive Summary
The Fecal Microbiota Transplantation market is experiencing robust growth, primarily driven by increasing cases of recurrent Clostridioides difficile infection (CDI) and expanding research into broader applications for gut dysbiosis-related conditions. Business trends indicate a shift towards the commercialization of standardized, regulated FMT products, moving beyond traditional clinical administration. Pharmaceutical and biotechnology companies are investing heavily in developing defined microbial consortia and encapsulated formulations to overcome the logistical and regulatory challenges associated with raw stool donations. This commercialization effort is supported by a growing number of regulatory approvals and guidelines, particularly in North America and Europe, fostering greater market access and patient confidence. Strategic partnerships between academic institutions, stool banks, and industry players are also shaping the competitive landscape, accelerating product development and market penetration.
Regional trends reveal North America as the dominant market, attributed to high CDI prevalence, advanced healthcare infrastructure, and significant research funding. Europe follows closely, with several countries establishing national guidelines and reimbursement policies for FMT. The Asia Pacific region is emerging as a high-growth market, driven by increasing healthcare expenditure, rising awareness, and a large patient population, though regulatory frameworks are still evolving. In terms of segments, the CDI indication continues to hold the largest market share due to its established efficacy and regulatory acceptance. However, the inflammatory bowel disease (IBD) and irritable bowel syndrome (IBS) segments are projected to witness the fastest growth, fueled by promising clinical trial results and a growing understanding of the microbiome's role in these chronic conditions. Furthermore, product segment trends show a gradual transition from traditional liquid FMT suspensions towards more patient-friendly, standardized oral capsule formulations and next-generation microbiome therapies, which offer improved convenience, safety, and scalability. This comprehensive evolution across business, regional, and segment trends underscores a dynamic and rapidly maturing market poised for significant expansion.
AI Impact Analysis on Fecal Microbiota Transplantation Market
Common user questions regarding AI's impact on the Fecal Microbiota Transplantation market frequently revolve around its potential to enhance donor screening accuracy, optimize patient selection, personalize treatment protocols, and accelerate the discovery of novel microbiome-based therapeutics. Users express curiosity about how AI can analyze complex microbiome data to identify optimal donor-recipient matches, predict treatment success, and even design synthetic microbial consortia, thereby overcoming current limitations related to donor variability and the complex regulatory landscape. Concerns often include data privacy, the reliability of AI algorithms in interpreting highly variable biological data, and the ethical implications of AI-driven medical decisions in such a novel therapeutic area. There is a clear expectation that AI will streamline research, improve clinical outcomes, and ultimately lead to more accessible and effective FMT therapies, transforming the field from an empirical procedure to a precision medicine approach.
- AI can significantly enhance donor screening by analyzing vast datasets of donor health records, lifestyle factors, and microbiome profiles to identify super-donors and mitigate transmission risks more effectively than current manual methods.
- Predictive analytics powered by AI can help in patient stratification, identifying which patients are most likely to respond positively to FMT for specific indications, thus optimizing treatment success rates and resource allocation.
- AI-driven bioinformatics tools can rapidly process and interpret complex metagenomic and metatranscriptomic data from patient and donor samples, leading to a deeper understanding of microbial dysbiosis and the mechanisms of FMT action.
- Machine learning algorithms can be employed in the design and optimization of synthetic microbial consortia, enabling the development of "designer" probiotics or defined communities that precisely target specific disease pathways, reducing reliance on human fecal material.
- AI platforms can accelerate drug discovery for microbiome-modulating therapies by identifying key microbial species or metabolic pathways correlated with disease states or treatment responses, opening new avenues for pharmaceutical intervention.
- Personalized treatment plans can be developed using AI to analyze an individual's unique microbiome, genetic makeup, and clinical history, tailoring the composition and timing of microbial interventions for optimal patient outcomes.
- Robotics and AI can automate the processing, quality control, and encapsulation of FMT products, leading to greater standardization, reduced labor costs, and improved scalability of therapeutic manufacturing.
DRO & Impact Forces Of Fecal Microbiota Transplantation Market
The Fecal Microbiota Transplantation (FMT) market is significantly influenced by a dynamic interplay of drivers, restraints, opportunities, and broader impact forces. Key drivers propelling market expansion include the escalating global incidence of recurrent Clostridioides difficile infection (CDI), which remains the most established and successful application of FMT, coupled with the growing challenge of antibiotic resistance necessitating alternative therapeutic strategies. Furthermore, increasing scientific understanding of the human gut microbiome's profound impact on health and disease, alongside a surge in research exploring FMT's efficacy across a spectrum of conditions like inflammatory bowel disease (IBD), irritable bowel syndrome (IBS), and various metabolic and neurological disorders, are broadening its addressable market and attracting substantial investment. Rising awareness among healthcare professionals and the public regarding microbiome health and the potential benefits of FMT further stimulates demand, while supportive regulatory developments in key regions are providing clearer pathways for commercialization and clinical adoption, enhancing market confidence and access.
However, several restraints pose challenges to the market's unbridled growth. These include significant regulatory hurdles, as authorities grapple with classifying FMT products (e.g., drug, biologic, or tissue), leading to fragmented and often stringent approval processes that can delay market entry for novel therapies. The complexities and costs associated with rigorous donor screening, ensuring safety and efficacy while minimizing potential pathogen transmission, also present a considerable operational and financial burden. Social stigma, ethical concerns surrounding the use of human fecal material, and a lack of standardized protocols across different clinics and countries contribute to hesitations in adoption. Furthermore, the absence of comprehensive long-term safety data for broader indications, and the inherent variability in donor material and treatment outcomes, require more robust research and consensus. Despite these challenges, significant opportunities exist for market players. These include the expansion of FMT applications into new, high-prevalence indications beyond CDI, particularly in chronic gastrointestinal and systemic diseases, leveraging the vast potential of microbiome modulation. The development of next-generation FMT products, such as encapsulated microbial consortia, synthetic microbial products, and rationally designed live biotherapeutic products, offers improved standardization, scalability, and patient convenience, overcoming many of the current limitations of raw stool. Global expansion into emerging markets, where healthcare infrastructure and awareness are rapidly improving, presents untapped patient populations. The overarching impact forces shaping the market include continuous technological advancements in microbiome analysis (e.g., next-generation sequencing, metabolomics), which are deepening scientific understanding and facilitating the development of targeted therapies. Changes in healthcare policies and reimbursement strategies, particularly as FMT gains more widespread acceptance and evidence, will be crucial in determining patient access and market penetration. Lastly, the escalating global burden of chronic gut-related diseases and antibiotic resistance continues to underscore the urgent need for innovative solutions like FMT, solidifying its long-term potential.
Segmentation Analysis
The Fecal Microbiota Transplantation market is intricately segmented to provide a comprehensive understanding of its diverse applications, product types, and end-user adoption patterns. This segmentation allows for targeted market strategies, identifying key growth areas and differentiating between various therapeutic approaches. The market is primarily categorized by indication, which reflects the specific health conditions FMT is used to treat; by product type, representing the different forms in which FMT is administered; by route of administration, detailing how the treatment is delivered to the patient; and by end-user, identifying the primary healthcare settings utilizing FMT therapies. Further segmentation by geographic region provides insights into varying regulatory landscapes, disease prevalence, and adoption rates across the globe, painting a holistic picture of market dynamics and potential for expansion in specific niches.
- By Indication
- Clostridioides difficile Infection (CDI)
- Inflammatory Bowel Disease (IBD)
- Irritable Bowel Syndrome (IBS)
- Metabolic Disorders (e.g., Obesity, Diabetes)
- Neurological Disorders (e.g., Autism, Parkinson's Disease)
- Other Indications (e.g., Graft-versus-host disease, Allergic diseases)
- By Product Type
- Liquid FMT
- Encapsulated FMT (Capsules)
- Stool Banks
- Defined Microbial Consortia / Synthetic FMT
- By Route of Administration
- Colonoscopy
- Endoscopy (Upper Endoscopy/Nasogastric Tube)
- Enema
- Oral Capsules
- By End User
- Hospitals
- Clinics
- Ambulatory Surgical Centers
- Research Institutions
Value Chain Analysis For Fecal Microbiota Transplantation Market
The value chain for the Fecal Microbiota Transplantation market is a multi-stage process involving rigorous screening, meticulous processing, and careful delivery of microbial therapeutics. It begins with the upstream activities of donor recruitment and stringent screening, which is a critical phase to ensure the safety and efficacy of the transplanted material. This involves extensive medical history reviews, serological testing for infectious diseases, and comprehensive stool sample analysis to exclude pathogens and identify healthy microbiome profiles. The reliability and quality of the donor pool directly impact the integrity of the entire value chain. Following selection, stool collection is performed, which then moves into the processing stage where the material is prepared for therapeutic use, often involving homogenization, filtration, and cryopreservation, particularly by dedicated stool banks or pharmaceutical companies developing standardized products.
Midstream activities primarily focus on the manufacturing, quality control, and storage of FMT products. Stool banks play a crucial role in preparing and storing liquid or encapsulated FMT preparations, adhering to strict quality and safety standards. For pharmaceutical companies developing next-generation live biotherapeutic products (LBPs) or defined microbial consortia, this stage involves complex fermentation, purification, and encapsulation processes under Good Manufacturing Practices (GMP). Quality control is paramount at every step to ensure the absence of pathogens, consistency of microbial composition, and product stability. Downstream activities encompass the distribution channel, which can be direct or indirect. Direct channels involve stool banks supplying FMT products directly to hospitals, clinics, or individual practitioners, often on a prescription basis. Indirect channels involve pharmaceutical companies marketing and distributing their approved FMT therapies through established healthcare distribution networks, including wholesalers and pharmacies, to reach hospitals and ambulatory surgical centers. The ultimate end-user in both scenarios remains the patient, with administration typically performed by gastroenterologists or other specialists. The efficiency and integrity of each stage, from donor identification to patient administration, are vital for the successful commercialization and broad adoption of FMT therapies, necessitating robust logistical support and regulatory oversight.
Fecal Microbiota Transplantation Market Potential Customers
The primary potential customers for Fecal Microbiota Transplantation (FMT) therapies are patients suffering from a range of gastrointestinal and systemic conditions linked to gut dysbiosis, particularly those who have exhausted conventional treatment options. At the forefront are individuals with recurrent Clostridioides difficile infection (CDI), especially those experiencing multiple relapses, as FMT has demonstrated exceptional efficacy in this patient group, often offering a definitive cure where antibiotics have failed. Beyond CDI, patients with moderate to severe inflammatory bowel disease (IBD), including Crohn's disease and ulcerative colitis, represent a significant and growing customer base, as ongoing research explores FMT's potential to induce and maintain remission by modulating the gut immune response and reducing inflammation. Similarly, individuals struggling with irritable bowel syndrome (IBS), characterized by chronic abdominal pain and altered bowel habits, are increasingly considered candidates for FMT as a novel approach to alleviate symptoms by restoring microbial balance.
Furthermore, the customer base extends to patients with various metabolic disorders such as obesity and type 2 diabetes, where gut microbiome alterations are implicated in disease pathogenesis, making them potential beneficiaries of FMT to improve metabolic health markers. Emerging research also identifies patients with certain neurological conditions, including autism spectrum disorders and Parkinson's disease, as prospective customers, as evidence linking the gut-brain axis to these conditions continues to mount. Beyond patients, hospitals, clinics, and ambulatory surgical centers represent crucial institutional customers, as they are the primary sites for administering FMT and purchasing approved therapies. Gastroenterologists, infectious disease specialists, and other clinicians specializing in gut health are key decision-makers and prescribers, influencing the adoption of FMT. Finally, academic and pharmaceutical research institutions are also significant consumers of FMT products and related services for ongoing clinical trials and the development of next-generation microbiome therapeutics. This broad spectrum of customers underscores the expansive and evolving market for FMT, driven by unmet medical needs and a growing understanding of the microbiome's therapeutic potential.
| Report Attributes | Report Details |
|---|---|
| Market Size in 2026 | $350 Million |
| Market Forecast in 2033 | $1,481.5 Million |
| Growth Rate | 22.5% CAGR |
| Historical Year | 2019 to 2024 |
| Base Year | 2025 |
| Forecast Year | 2026 - 2033 |
| DRO & Impact Forces |
|
| Segments Covered |
|
| Key Companies Covered | Seres Therapeutics, Rebiotix (Ferring Pharmaceuticals), Vedanta Biosciences, Finch Therapeutics, MaaT Pharma, OpenBiome, Enterome, Evelo Biosciences, Takeda Pharmaceutical Company Limited, Pendulum Therapeutics, Microbiotica, 4D pharma plc, BiomX, EnteroBiotix, Caelus Health, Kallyope Inc., TargEDys, Daicel Corporation, ADM Biopolis, Symbiotix. |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Enquiry Before Buy | Have specific requirements? Send us your enquiry before purchase to get customized research options. Request For Enquiry Before Buy |
Fecal Microbiota Transplantation Market Key Technology Landscape
The Fecal Microbiota Transplantation market is underpinned by a rapidly evolving technological landscape that drives innovation in donor screening, microbial analysis, product formulation, and delivery methods. Core to this landscape are advanced molecular diagnostics, particularly next-generation sequencing (NGS) and quantitative polymerase chain reaction (qPCR), which enable highly detailed and accurate profiling of donor and recipient microbiomes. These technologies are crucial for identifying diverse microbial communities, detecting potential pathogens, and assessing the overall health and stability of donor stool. Beyond basic sequencing, advanced bioinformatics and computational biology play a vital role in processing the enormous amounts of data generated, allowing for deeper insights into microbial interactions, metabolic pathways, and the identification of key species or functional groups relevant to disease or therapeutic efficacy. This analytical power is essential for both understanding FMT mechanisms and for developing highly targeted, next-generation microbiome therapeutics.
Furthermore, the development of sophisticated cryopreservation techniques is critical for maintaining the viability and diversity of microbial communities in stool samples during storage and transportation, ensuring product integrity for stool banks and commercial suppliers. Aseptic processing and manufacturing technologies, operating under stringent Good Manufacturing Practice (GMP) standards, are paramount for producing safe, consistent, and scalable FMT products, particularly as the industry moves towards regulated, commercialized therapies. This includes the development of specialized encapsulation technologies, such as enteric-coated capsules, which protect the microbial content from gastric acid and ensure targeted delivery to the lower gastrointestinal tract, significantly improving patient convenience and treatment adherence. Beyond direct FMT, advancements in synthetic biology and microbial engineering are paving the way for defined microbial consortia, where specific, beneficial bacteria are cultured and combined to create a "designer" microbiome product. This approach promises greater standardization, reduced safety risks associated with whole stool, and the ability to precisely tailor treatments to individual patient needs. The integration of these diverse technologies, from high-throughput sequencing to advanced manufacturing and synthetic biology, is collectively transforming FMT from an empirical procedure into a sophisticated, precision medicine approach, poised to unlock new therapeutic frontiers.
Regional Highlights
- North America: This region consistently dominates the Fecal Microbiota Transplantation market, primarily driven by a high prevalence of recurrent Clostridioides difficile infection (CDI) and a well-established healthcare infrastructure that supports advanced medical procedures. The presence of leading research institutions, significant government and private funding for microbiome research, and relatively clear, albeit evolving, regulatory pathways for FMT products contribute substantially to market growth. Strong awareness among both healthcare providers and the patient population regarding the benefits of FMT, coupled with favorable reimbursement policies, further solidifies its leading position. The United States, in particular, has seen several FDA-approved or commercially available FMT products, setting a global precedent for market development and therapeutic adoption.
- Europe: The European market for FMT is robust, characterized by a strong academic research base, particularly in countries like the UK, Germany, and France, which are actively investigating new indications beyond CDI. While regulatory landscapes vary across individual European countries, the European Medicines Agency (EMA) and national health authorities are progressively establishing guidelines for the use and commercialization of FMT, fostering a more standardized approach. Growing public and medical professional awareness of the gut microbiome's role in health, coupled with increasing investments in biotechnology and pharmaceutical sectors focused on live biotherapeutic products, fuels the demand for FMT therapies. The availability of national stool banks and a rising incidence of chronic gastrointestinal diseases also contribute to the region's steady market expansion.
- Asia Pacific (APAC): The APAC region represents the fastest-growing market for Fecal Microbiota Transplantation, driven by several powerful factors including a rapidly improving healthcare infrastructure, increasing healthcare expenditure, and a large patient population susceptible to gastrointestinal disorders. Countries such as China, India, Japan, and South Korea are witnessing a surge in research activities related to the microbiome and FMT, along with a growing interest from local pharmaceutical and biotech companies. While regulatory frameworks are still maturing in many parts of the region, rising awareness among medical professionals and patients, coupled with the increasing prevalence of lifestyle-related diseases, creates significant untapped potential. Economic development and greater access to advanced medical treatments are key accelerators for market expansion across diverse APAC economies.
- Latin America: The FMT market in Latin America is currently in its nascent stage but holds considerable growth potential. Factors contributing to this growth include improving healthcare access, increasing investment in healthcare infrastructure, and a rising awareness among medical practitioners regarding advanced treatment options for gastrointestinal conditions. Countries like Brazil and Mexico are leading the adoption of FMT, primarily for recurrent CDI, driven by local research initiatives and a gradual shift towards evidence-based medicine. However, challenges such as limited regulatory clarity, economic disparities, and lower public awareness compared to developed regions persist. As healthcare systems mature and the burden of chronic diseases rises, Latin America is expected to become a more prominent market for FMT.
- Middle East and Africa (MEA): The MEA region presents a developing market for Fecal Microbiota Transplantation. Growth drivers include increasing prevalence of gastrointestinal infections and chronic diseases, coupled with growing healthcare expenditure and efforts to modernize medical facilities, particularly in countries within the Gulf Cooperation Council (GCC). Research and clinical interest in FMT are emerging, but the market faces significant hurdles such as varying regulatory environments, cultural sensitivities surrounding the procedure, and a general lack of widespread awareness and infrastructure for specialized microbiome therapies. Collaborative efforts with international research bodies and the establishment of local expertise will be critical for accelerating market penetration and patient access in this region.
Top Key Players
The market research report includes a detailed profile of leading stakeholders in the Fecal Microbiota Transplantation Market.- Seres Therapeutics
- Rebiotix (a Ferring Pharmaceuticals Company)
- Vedanta Biosciences
- Finch Therapeutics
- MaaT Pharma
- OpenBiome
- Enterome
- Evelo Biosciences
- Takeda Pharmaceutical Company Limited
- Pendulum Therapeutics
- Microbiotica
- 4D pharma plc
- BiomX
- EnteroBiotix
- Caelus Health
- Kallyope Inc.
- TargEDys
- Daicel Corporation
- ADM Biopolis
- Symbiotix
Frequently Asked Questions
Analyze common user questions about the Fecal Microbiota Transplantation market and generate a concise list of summarized FAQs reflecting key topics and concerns.What is Fecal Microbiota Transplantation (FMT) and how does it work?
Fecal Microbiota Transplantation (FMT) is a procedure where fecal matter from a healthy donor is transferred into the gastrointestinal tract of a recipient patient. The objective is to restore a balanced and healthy gut microbiome, particularly when the patient's own microbial community has been disrupted, often by antibiotics. The diverse microorganisms from the donor stool colonize the recipient's gut, outcompeting harmful bacteria and re-establishing beneficial functions, such as nutrient absorption and immune modulation. This process effectively resets the gut environment to a healthier state, allowing the patient's body to recover from dysbiosis-related conditions.
What conditions can Fecal Microbiota Transplantation treat?
The most established and FDA-approved indication for Fecal Microbiota Transplantation (FMT) is recurrent Clostridioides difficile infection (CDI), especially when standard antibiotic treatments have failed. Beyond CDI, significant clinical research is exploring FMT's potential in treating various other conditions associated with gut dysbiosis, including inflammatory bowel disease (IBD) such as Crohn's disease and ulcerative colitis, irritable bowel syndrome (IBS), and certain metabolic disorders. Early studies also show promise in neurological conditions and autoimmune diseases, although these applications are still largely experimental and under investigation, requiring further rigorous clinical trials to establish efficacy and safety for widespread use.
What are the potential risks and side effects of FMT?
While Fecal Microbiota Transplantation (FMT) is generally considered safe, particularly for recurrent CDI, potential risks and side effects exist. Common side effects are usually mild and transient, including temporary abdominal discomfort, bloating, flatulence, and diarrhea immediately following the procedure. More serious, albeit rare, risks include the theoretical transmission of infectious agents (bacteria, viruses, parasites) from the donor, despite rigorous screening processes. There is also a small risk of complications related to the administration method, such as perforation during colonoscopy. Long-term safety data for non-CDI indications are still being collected, and the potential for altering the recipient's microbiome in unforeseen ways is a subject of ongoing research and careful monitoring.
Are FMT products regulated, and how are they administered?
The regulation of Fecal Microbiota Transplantation (FMT) products varies significantly by region. In the United States, the FDA generally regulates FMT as a drug or biologic, requiring investigational new drug (IND) applications for most uses outside of recurrent C. difficile infection unresponsive to standard therapies. In Europe, national health authorities and the European Medicines Agency (EMA) are developing guidelines, with some countries classifying it as a tissue or blood product. Administration methods also vary; common routes include colonoscopy or enema for direct delivery to the colon, upper endoscopy or nasogastric tube for delivery to the upper GI tract, and increasingly, oral capsules which offer a less invasive and more patient-friendly option for standardized commercial products. The choice of administration method often depends on the specific indication, product formulation, and clinical judgment.
What is the future outlook for the Fecal Microbiota Transplantation market?
The future outlook for the Fecal Microbiota Transplantation market is highly promising, with significant growth anticipated driven by expanding indications beyond recurrent C. difficile infection and the development of next-generation therapies. The market is expected to shift towards more standardized, encapsulated, and synthetic microbial products, offering improved safety, consistency, and patient convenience compared to traditional fresh stool preparations. Advances in microbiome science, including AI-driven analytics and synthetic biology, will enable the creation of highly targeted and personalized microbiome therapeutics. Increased investment from pharmaceutical companies, clearer regulatory pathways, and rising global awareness of gut health are poised to transform FMT into a mainstream therapeutic modality for a wide array of chronic diseases, unlocking substantial market potential.
To check our Table of Contents, please mail us at: sales@marketresearchupdate.com
Research Methodology
The Market Research Update offers technology-driven solutions and its full integration in the research process to be skilled at every step. We use diverse assets to produce the best results for our clients. The success of a research project is completely reliant on the research process adopted by the company. Market Research Update assists its clients to recognize opportunities by examining the global market and offering economic insights. We are proud of our extensive coverage that encompasses the understanding of numerous major industry domains.
Market Research Update provide consistency in our research report, also we provide on the part of the analysis of forecast across a gamut of coverage geographies and coverage. The research teams carry out primary and secondary research to implement and design the data collection procedure. The research team then analyzes data about the latest trends and major issues in reference to each industry and country. This helps to determine the anticipated market-related procedures in the future. The company offers technology-driven solutions and its full incorporation in the research method to be skilled at each step.
The Company's Research Process Has the Following Advantages:
- Information Procurement
The step comprises the procurement of market-related information or data via different methodologies & sources.
- Information Investigation
This step comprises the mapping and investigation of all the information procured from the earlier step. It also includes the analysis of data differences observed across numerous data sources.
- Highly Authentic Source
We offer highly authentic information from numerous sources. To fulfills the client’s requirement.
- Market Formulation
This step entails the placement of data points at suitable market spaces in an effort to assume possible conclusions. Analyst viewpoint and subject matter specialist based examining the form of market sizing also plays an essential role in this step.
- Validation & Publishing of Information
Validation is a significant step in the procedure. Validation via an intricately designed procedure assists us to conclude data-points to be used for final calculations.
×
Request Free Sample:
Related Reports
Select License

Why Choose Us

We're cost-effective and Offered Best services:
We are flexible and responsive startup research firm. We adapt as your research requires change, with cost-effectiveness and highly researched report that larger companies can't match.

Information Safety
Market Research Update ensure that we deliver best reports. We care about the confidential and personal information quality, safety, of reports. We use Authorize secure payment process.

We Are Committed to Quality and Deadlines
We offer quality of reports within deadlines. We've worked hard to find the best ways to offer our customers results-oriented and process driven consulting services.

Our Remarkable Track Record
We concentrate on developing lasting and strong client relationship. At present, we hold numerous preferred relationships with industry leading firms that have relied on us constantly for their research requirements.

Best Service Assured
Buy reports from our executives that best suits your need and helps you stay ahead of the competition.

Customized Research Reports
Our research services are custom-made especially to you and your firm in order to discover practical growth recommendations and strategies. We don't stick to a one size fits all strategy. We appreciate that your business has particular research necessities.

Service Assurance
At Market Research Update, we are dedicated to offer the best probable recommendations and service to all our clients. You will be able to speak to experienced analyst who will be aware of your research requirements precisely.
Contact With Our Sales Team
Customer Testimonials
The content of the report is always up to the mark. Good to see speakers from expertise authorities.
Privacy requested , Managing Director
A lot of unique and interesting topics which are described in good manner.
Privacy requested, President
Well researched, expertise analysts, well organized, concrete and current topics delivered in time.
Privacy requested, Development Manager