Human Rotavirus Vaccine Market Size, By Region (North America, Europe, Asia-Pacific, Latin America, Middle East and Africa), By Statistics, Trends, Outlook and Forecast 2026 to 2033 (Financial Impact Analysis)
ID : MRU_ 444126 | Date : Feb, 2026 | Pages : 258 | Region : Global | Publisher : MRU
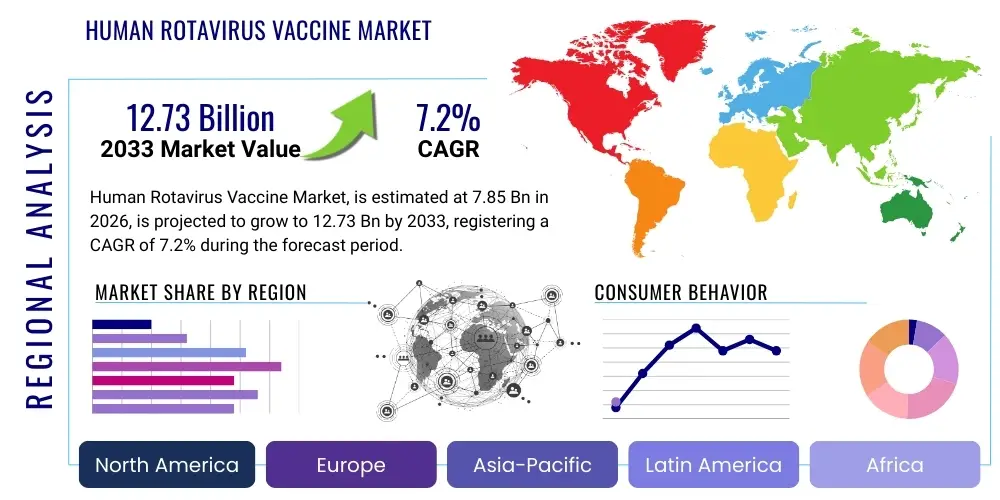
Human Rotavirus Vaccine Market Size
The Human Rotavirus Vaccine Market is projected to grow at a Compound Annual Growth Rate (CAGR) of 7.2% between 2026 and 2033. The market is estimated at USD 7.85 billion in 2026 and is projected to reach USD 12.73 billion by the end of the forecast period in 2033. This growth is primarily driven by the increasing global emphasis on infant immunization programs, the high burden of rotavirus disease in developing countries, and continuous efforts by public health organizations to expand vaccine access and affordability.
Human Rotavirus Vaccine Market introduction
The Human Rotavirus Vaccine Market is a critical segment within the global pharmaceuticals and biotechnology industry, dedicated to preventing rotavirus infections, which are the leading cause of severe diarrheal disease in infants and young children worldwide. Rotavirus vaccines are typically live-attenuated oral vaccines designed to elicit an immune response that protects against the various strains of the virus. The primary products dominating this market include Rotarix (manufactured by GlaxoSmithKline) and RotaTeq (manufactured by Merck & Co., Inc.), alongside regionally significant vaccines like Rotavac (Bharat Biotech) and Rotasiil (Serum Institute of India).
Major applications of these vaccines lie in universal infant immunization programs implemented by national governments and supported by international organizations such as the World Health Organization (WHO) and Gavi, the Vaccine Alliance. These programs aim to reduce child mortality and morbidity associated with rotavirus gastroenteritis. The benefits of widespread rotavirus vaccination are profound, including a significant reduction in hospitalizations and deaths due to severe diarrhea, decreased healthcare expenditures, and improved overall child health outcomes, particularly in low-income settings where access to medical care for diarrheal diseases can be limited.
Key driving factors for market expansion include the persistent global burden of rotavirus disease, especially in regions with poor sanitation and limited access to healthcare, leading to high demand for effective preventive measures. Furthermore, robust support from global health initiatives and governmental policies advocating for mandatory or recommended infant immunization schedules significantly bolsters market growth. Continuous research and development efforts to improve vaccine efficacy, expand strain coverage, and enhance vaccine stability for easier distribution in challenging environments also contribute to the market's positive trajectory. Public awareness campaigns regarding the importance of early childhood vaccination play a crucial role in improving vaccine uptake rates.
Human Rotavirus Vaccine Market Executive Summary
The Human Rotavirus Vaccine Market is experiencing robust growth, primarily fueled by global public health initiatives and the undeniable efficacy of these vaccines in reducing severe diarrheal disease among infants. Business trends indicate a strategic focus on expanding manufacturing capacities and distribution networks, especially in emerging economies, where the disease burden is highest. Pharmaceutical companies are increasingly collaborating with non-profit organizations and governments to facilitate vaccine procurement and ensure equitable access, reflecting a shift towards public-private partnerships as a core business model. Additionally, there is an ongoing emphasis on developing more cost-effective vaccines and optimizing cold chain logistics to overcome accessibility challenges in remote areas, which is critical for market penetration and sustained growth.
Regional trends highlight the significant impact of national immunization programs. Asia Pacific and Africa represent key growth epicenters due to their large birth cohorts, high prevalence of rotavirus infection, and increasing government investments in child health. While mature markets in North America and Europe maintain stable demand driven by established universal immunization schedules, their growth is primarily in product innovation and premium vaccine offerings. Latin America demonstrates a steady upward trend, benefiting from strong governmental commitment to immunization. The Middle East is also showing promising growth as healthcare infrastructure improves and vaccination programs expand.
Segment trends indicate that live-attenuated oral vaccines will continue to dominate the market, with product differentiation focusing on serotype coverage, ease of administration, and storage stability. The market sees a division between multinational pharmaceutical giants offering globally recognized brands and regional players providing more affordable options tailored for specific local markets. Demand from public health programs and governmental procurement agencies forms the largest segment of end-users, reflecting the public good nature of these vaccines. However, private clinics and hospitals also contribute significantly, particularly in regions where out-of-pocket payment for vaccinations is common or public supply is limited, thus creating diverse revenue streams across the value chain.
AI Impact Analysis on Human Rotavirus Vaccine Market
Users are keen to understand how artificial intelligence can revolutionize vaccine development, deployment, and public health outcomes within the Human Rotavirus Vaccine Market. Key themes emerging from common user questions revolve around AI's potential to accelerate the discovery of novel vaccine antigens, optimize manufacturing processes for enhanced efficiency and cost-effectiveness, and improve real-time surveillance of disease outbreaks and vaccine effectiveness. There is also significant interest in AI's role in predicting future viral strains, personalizing vaccination strategies, and streamlining complex cold chain logistics. Concerns often touch upon data privacy, the ethical implications of AI-driven health interventions, and the need for robust validation to ensure AI algorithms contribute positively and reliably to public health without introducing new risks or biases.
- AI can accelerate vaccine discovery by analyzing vast genomic and proteomic datasets to identify potential vaccine candidates and predict their immunogenicity and cross-protectivity against various rotavirus strains.
- Manufacturing processes can be optimized through AI-driven predictive maintenance, quality control, and process parameter adjustments, leading to higher yields, reduced waste, and lower production costs for rotavirus vaccines.
- AI enhances epidemiological surveillance by analyzing real-time health data, social media trends, and environmental factors to predict rotavirus outbreaks, enabling proactive public health interventions and targeted vaccination campaigns.
- Improved cold chain management is facilitated by AI algorithms that monitor temperature, humidity, and location of vaccine shipments, flagging potential deviations and optimizing delivery routes to ensure vaccine potency and reduce spoilage, especially in remote areas.
- AI supports personalized vaccination schedules by leveraging individual health data, genetic predispositions, and local epidemiological patterns to recommend optimal timing and type of rotavirus vaccine for infants, though this is still largely theoretical for current products.
- Public health communication and addressing vaccine hesitancy can be bolstered by AI-powered tools that analyze public sentiment, identify misinformation, and tailor educational messages to specific demographic groups, promoting vaccine acceptance.
DRO & Impact Forces Of Human Rotavirus Vaccine Market
The Human Rotavirus Vaccine Market is significantly shaped by a dynamic interplay of drivers, restraints, opportunities, and external impact forces. A primary driver is the substantial global burden of rotavirus disease, which continues to cause hundreds of thousands of deaths annually among young children, particularly in low-income countries, thereby creating an imperative for effective preventive measures. Government mandates and robust support for universal immunization programs from international bodies like the WHO, UNICEF, and Gavi further accelerate vaccine uptake. Additionally, continuous advancements in vaccine research and development, aiming for broader serotype coverage and improved stability, contribute positively to market expansion. The growing public and parental awareness regarding the benefits of early childhood vaccination also plays a crucial role in driving demand. These factors collectively create a strong foundation for sustained market growth.
However, several significant restraints impede the market's full potential. The relatively high cost of rotavirus vaccines, while decreasing with volume procurement for public programs, remains a barrier for private markets in many regions. The strict cold chain requirements for transportation and storage of live-attenuated vaccines pose logistical challenges, particularly in rural and underserved areas lacking adequate infrastructure. Vaccine hesitancy, fueled by misinformation and lack of trust in healthcare systems, continues to affect uptake rates in various communities globally. Furthermore, the limited number of manufacturers for globally recognized brands can create supply chain vulnerabilities, while the need for multiple doses for full protection can sometimes lead to incomplete vaccination schedules for infants.
Despite these challenges, numerous opportunities exist for market growth and innovation. The expansion of national immunization programs into previously underserved regions, especially in parts of Asia and Africa with high birth rates and disease incidence, presents a significant untapped market. Development of new vaccine formulations that offer improved thermal stability or single-dose efficacy could revolutionize distribution and compliance. Exploring combination vaccines that include rotavirus components could simplify immunization schedules and enhance coverage. Additionally, leveraging advanced manufacturing techniques to reduce production costs and increase supply volume offers a pathway to greater affordability and accessibility. Strategic partnerships between pharmaceutical companies, governments, and NGOs are vital to navigate these opportunities effectively, ensuring broader reach and impact. The continuous monitoring of evolving rotavirus strains also presents an opportunity for targeted vaccine development, maintaining efficacy in the face of viral evolution. The ongoing digital transformation in healthcare also opens avenues for improved data collection on vaccine efficacy and coverage, allowing for more data-driven public health strategies and rapid responses to epidemiological shifts.
Segmentation Analysis
The Human Rotavirus Vaccine Market is comprehensively segmented to provide a detailed understanding of its various components and underlying dynamics. This segmentation allows for targeted strategies, resource allocation, and a clearer view of market opportunities and challenges across different product types, end-user categories, and distribution channels. The market's structure reflects the diverse approaches to vaccine development, delivery, and administration globally, catering to both public health imperatives and private healthcare needs. Understanding these segments is crucial for stakeholders to identify specific growth areas and tailor their offerings to meet the unique demands of each market niche.
- By Product Type
- Rotarix (GlaxoSmithKline)
- RotaTeq (Merck & Co., Inc.)
- Rotavac (Bharat Biotech)
- Rotasiil (Serum Institute of India)
- Other Live-Attenuated Oral Vaccines
- By End-User
- Hospitals
- Clinics
- Public Health Programs
- Government & NGOs
- Research Institutes
- By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Government Procurement & Supply Channels
- Online Pharmacies
- Direct Sales
- By Age Group
- Infants (6 weeks to 6 months)
- Children (Above 6 months - Not primary target, but related considerations)
Value Chain Analysis For Human Rotavirus Vaccine Market
The value chain for the Human Rotavirus Vaccine Market encompasses a series of interconnected activities, beginning from upstream research and development through to downstream distribution and post-market surveillance. Upstream activities are critical, involving extensive biological research, preclinical studies, and clinical trials to identify, develop, and validate vaccine candidates. This phase typically includes partnerships between academic institutions, biotechnology firms, and large pharmaceutical companies, focusing on virus strain identification, attenuation techniques, and formulation development. The quality and safety benchmarks established during this stage are paramount for regulatory approval and market acceptance.
The midstream segment of the value chain focuses on the complex manufacturing processes, including cell culture, viral propagation, purification, and formulation, followed by sterile filling and packaging. This stage requires significant capital investment in specialized facilities that adhere to stringent Good Manufacturing Practices (GMP) to ensure vaccine potency and safety. Logistics and supply chain management play a crucial role here, especially the maintenance of a robust cold chain for temperature-sensitive live-attenuated vaccines, which must be preserved from the point of manufacture through to the point of administration. This involves specialized warehousing, transportation, and monitoring systems.
Downstream activities involve the distribution channel, which is often bifurcated into direct and indirect routes. Direct channels include sales to national governments and large international organizations (like UNICEF, Gavi) for public immunization programs, often involving large volume tenders and highly coordinated logistics. Indirect channels involve sales to wholesalers, distributors, hospital pharmacies, and retail pharmacies that serve private healthcare providers and individual consumers. Both channels require careful management to ensure vaccine availability, affordability, and proper handling. Post-market surveillance, pharmacovigilance, and continuous public health communication are also integral to the downstream process, monitoring vaccine safety and effectiveness in real-world settings and maintaining public trust. This comprehensive value chain ensures that the vaccines reach the target population effectively and safely, contributing to global health outcomes.
Human Rotavirus Vaccine Market Potential Customers
The primary potential customers and end-users of human rotavirus vaccines are predominantly infants and young children, making parents, guardians, and pediatric healthcare providers key decision-makers and influencers. The ultimate recipients are infants typically between 6 weeks and 6 months of age, in accordance with recommended vaccination schedules designed to provide protection before exposure to the virus. However, the purchasing entities are more diverse, spanning both public and private sectors, reflecting the global strategies for vaccine distribution and access.
Governments, through their national immunization programs, represent the largest customer segment. They procure vast quantities of rotavirus vaccines via tender processes or direct agreements with manufacturers. These vaccines are then distributed free or at highly subsidized rates through public health clinics, hospitals, and community vaccination drives, aiming for universal coverage to achieve herd immunity and reduce disease burden at a population level. International non-governmental organizations (NGOs) such as Gavi, the Vaccine Alliance, and UNICEF also act as significant purchasers and facilitators, particularly for low-income countries, by negotiating favorable pricing and supporting distribution infrastructure to ensure equitable access.
Within the private sector, potential customers include private hospitals, pediatric clinics, and individual practitioners who offer vaccination services to patients willing to pay out-of-pocket or through private health insurance. Retail pharmacies also serve as indirect customers by stocking vaccines for prescription fulfillment. In these settings, parents and guardians are the direct buyers, often influenced by recommendations from their pediatricians. These various customer segments highlight the dual nature of the rotavirus vaccine market, catering to both public health mandates and individual healthcare choices, underscoring the necessity for differentiated marketing and distribution strategies across regions and socioeconomic strata.
| Report Attributes | Report Details |
|---|---|
| Market Size in 2026 | USD 7.85 Billion |
| Market Forecast in 2033 | USD 12.73 Billion |
| Growth Rate | 7.2% CAGR |
| Historical Year | 2019 to 2024 |
| Base Year | 2025 |
| Forecast Year | 2026 - 2033 |
| DRO & Impact Forces |
|
| Segments Covered |
|
| Key Companies Covered | GlaxoSmithKline plc (GSK), Merck & Co., Inc., Bharat Biotech International Limited, Serum Institute of India Pvt. Ltd., Lanzhou Institute of Biological Products Co., Ltd., Sanofi (through its acquisition of Shantha Biotechnics), Chengdu Institute of Biological Products Co., Ltd., Astellas Pharma Inc., Johnson & Johnson, Pfizer Inc., Valneva SE, Crucell (J&J), SK Bioscience Co. Ltd., Mitsubishi Tanabe Pharma Corporation, Takeda Pharmaceutical Company Limited, CureVac N.V. |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Enquiry Before Buy | Have specific requirements? Send us your enquiry before purchase to get customized research options. Request For Enquiry Before Buy |
Human Rotavirus Vaccine Market Key Technology Landscape
The key technology landscape underpinning the Human Rotavirus Vaccine Market primarily revolves around the sophisticated methods required for developing and producing live-attenuated oral vaccines. The core technology involves the isolation and attenuation of rotavirus strains to reduce their pathogenicity while retaining their immunogenicity. This is typically achieved through repeated passaging in cell cultures or animal models, a process that gradually diminishes the virus's ability to cause disease but allows it to replicate sufficiently in the host to elicit a protective immune response. Critical to this process are robust cell culture techniques, often utilizing Vero cells or other mammalian cell lines, for high-yield viral propagation.
Beyond the initial attenuation, advanced purification and formulation technologies are crucial to ensure vaccine safety, stability, and efficacy. Filtration, chromatography, and ultrafiltration techniques are employed to purify the attenuated virus particles from cell culture components, ensuring a highly purified and safe product. Formulation science focuses on creating stable liquid or lyophilized (freeze-dried) vaccine presentations that can withstand varied storage conditions, especially important for distribution in regions with limited cold chain infrastructure. Excipients, such as buffers, stabilizers, and cryoprotectants, are carefully selected to maintain viral viability over the vaccine's shelf life.
Furthermore, the development of multivalent vaccines, which protect against multiple rotavirus serotypes, represents a significant technological advancement. This involves combining several attenuated viral strains into a single vaccine, requiring careful balancing of immunogenicity and compatibility of each strain. Technologies aimed at improving thermal stability, such as novel lyophilization processes or specialized packaging, are continuously being explored to reduce reliance on strict cold chains, thereby enhancing accessibility in challenging environments. The ongoing application of genomics and proteomics in vaccine design also contributes to a deeper understanding of rotavirus biology and the identification of potential targets for next-generation vaccines, though current market products are predominantly based on established live-attenuated platforms.
Regional Highlights
- North America: This region, comprising the United States and Canada, represents a mature market with high vaccine coverage rates for rotavirus. Universal immunization programs are well-established, contributing to significantly reduced rates of rotavirus-associated gastroenteritis and hospitalizations. Market growth here is primarily driven by consistent birth rates, sustained public health awareness, and the continued adoption of newer vaccine formulations. The region also plays a crucial role in vaccine research and development, setting benchmarks for regulatory standards and innovation.
- Europe: Similar to North America, European countries generally have well-structured national immunization programs, with high uptake of rotavirus vaccines. Western European nations exhibit strong market penetration, while Eastern European countries are gradually expanding their coverage. The market benefits from robust healthcare infrastructure, government funding for public health, and increasing public acceptance of childhood vaccination. Variations exist across countries regarding specific vaccine recommendations and procurement policies, but the overall trend is towards increased coverage.
- Asia Pacific (APAC): The APAC region is a high-growth market, driven by its vast population, high birth rates, and a significant burden of rotavirus disease. Countries like India, China, Indonesia, and Vietnam are either implementing or expanding their national immunization programs, leading to substantial demand for rotavirus vaccines. Local manufacturers play a vital role in providing affordable vaccines, often with support from international organizations. Improvements in healthcare infrastructure and rising public awareness about preventable diseases are key drivers for market expansion in this region, despite challenges related to diverse healthcare systems and socio-economic disparities.
- Latin America: This region has shown strong commitment to rotavirus vaccination, with many countries having successfully integrated vaccines into their national immunization schedules. Mexico, Brazil, Argentina, and Colombia are prominent markets, demonstrating high vaccination rates and positive public health outcomes. Government procurement forms the backbone of the market, ensuring broad access. Continued efforts to reach underserved populations and maintain high coverage are central to market dynamics here.
- Middle East and Africa (MEA): The MEA region represents a market with immense growth potential, characterized by a substantial unmet need for rotavirus vaccination and a high incidence of diarrheal diseases. While some countries in the Middle East have established robust immunization programs, many African nations are in the nascent stages of implementing widespread rotavirus vaccination, often heavily reliant on international aid and organizations like Gavi. Improvements in healthcare funding, infrastructure development, and sustained efforts to combat vaccine-preventable diseases are crucial for unlocking the full market potential in this diverse and challenging region.
Top Key Players
The market research report includes a detailed profile of leading stakeholders in the Human Rotavirus Vaccine Market.- GlaxoSmithKline plc (GSK)
- Merck & Co., Inc.
- Bharat Biotech International Limited
- Serum Institute of India Pvt. Ltd.
- Lanzhou Institute of Biological Products Co., Ltd.
- Sanofi (through its acquisition of Shantha Biotechnics)
- Chengdu Institute of Biological Products Co., Ltd.
- Astellas Pharma Inc.
- Johnson & Johnson
- Pfizer Inc.
- Valneva SE
- Crucell (J&J)
- SK Bioscience Co. Ltd.
- Mitsubishi Tanabe Pharma Corporation
- Takeda Pharmaceutical Company Limited
- CureVac N.V.
- CanSino Biologics Inc.
- Panacea Biotec Ltd.
- Butantan Institute
- Novartis AG
Frequently Asked Questions
Analyze common user questions about the Human Rotavirus Vaccine market and generate a concise list of summarized FAQs reflecting key topics and concerns.What is the Human Rotavirus Vaccine and why is it important for infants?
The Human Rotavirus Vaccine is an oral vaccine administered to infants to prevent severe gastroenteritis caused by rotavirus, a leading cause of severe diarrhea, vomiting, fever, and abdominal pain in young children worldwide. It is crucial because rotavirus infections can lead to severe dehydration, hospitalization, and even death, particularly in developing countries with limited access to healthcare. Vaccination significantly reduces this burden, protecting infant health and alleviating pressure on healthcare systems.
What are the primary types of rotavirus vaccines available in the market?
Currently, the primary types of rotavirus vaccines globally are live-attenuated oral vaccines, notably Rotarix (GlaxoSmithKline) and RotaTeq (Merck & Co., Inc.). Rotarix is a monovalent vaccine targeting one major rotavirus strain, while RotaTeq is pentavalent, providing protection against five major strains. Regionally, other significant vaccines include Rotavac (Bharat Biotech) and Rotasiil (Serum Institute of India), which are also live-attenuated oral formulations developed for specific local epidemiological needs and affordability.
What are the common side effects and safety considerations for rotavirus vaccines?
Rotavirus vaccines are generally safe and well-tolerated. Common side effects are usually mild and temporary, including fussiness, mild diarrhea, and vomiting. A rare but serious side effect is intussusception, a type of bowel obstruction, which has been reported in very few cases, primarily within a week of the first dose. Healthcare providers thoroughly weigh the small risk of intussusception against the substantial benefits of preventing severe rotavirus disease, which is far more common and dangerous.
How do global immunization programs and government policies influence the rotavirus vaccine market?
Global immunization programs and government policies are paramount drivers for the rotavirus vaccine market. Initiatives by organizations like WHO, Gavi, and UNICEF, along with national government mandates for universal infant vaccination, significantly boost demand. These programs often involve large-scale procurement, distribution, and administration, ensuring broad access and affordability, especially in low-income countries where the disease burden is highest. Policies supporting mandatory vaccination, funding for research, and cold chain infrastructure are critical for market growth and public health impact.
What technological advancements are shaping the future of rotavirus vaccine development?
Future rotavirus vaccine development is being shaped by efforts to enhance thermal stability, potentially leading to vaccines that do not require strict cold chain storage, simplifying distribution in remote areas. Research also focuses on broadening serotype coverage to protect against emerging strains and exploring novel delivery methods or formulations, such as combination vaccines, to streamline infant immunization schedules. Additionally, advanced analytics and AI are being explored to optimize manufacturing, predict outbreaks, and improve surveillance, though these are nascent applications for existing products.
To check our Table of Contents, please mail us at: sales@marketresearchupdate.com
Research Methodology
The Market Research Update offers technology-driven solutions and its full integration in the research process to be skilled at every step. We use diverse assets to produce the best results for our clients. The success of a research project is completely reliant on the research process adopted by the company. Market Research Update assists its clients to recognize opportunities by examining the global market and offering economic insights. We are proud of our extensive coverage that encompasses the understanding of numerous major industry domains.
Market Research Update provide consistency in our research report, also we provide on the part of the analysis of forecast across a gamut of coverage geographies and coverage. The research teams carry out primary and secondary research to implement and design the data collection procedure. The research team then analyzes data about the latest trends and major issues in reference to each industry and country. This helps to determine the anticipated market-related procedures in the future. The company offers technology-driven solutions and its full incorporation in the research method to be skilled at each step.
The Company's Research Process Has the Following Advantages:
- Information Procurement
The step comprises the procurement of market-related information or data via different methodologies & sources.
- Information Investigation
This step comprises the mapping and investigation of all the information procured from the earlier step. It also includes the analysis of data differences observed across numerous data sources.
- Highly Authentic Source
We offer highly authentic information from numerous sources. To fulfills the client’s requirement.
- Market Formulation
This step entails the placement of data points at suitable market spaces in an effort to assume possible conclusions. Analyst viewpoint and subject matter specialist based examining the form of market sizing also plays an essential role in this step.
- Validation & Publishing of Information
Validation is a significant step in the procedure. Validation via an intricately designed procedure assists us to conclude data-points to be used for final calculations.
×
Request Free Sample:
Related Reports
Select License

Why Choose Us

We're cost-effective and Offered Best services:
We are flexible and responsive startup research firm. We adapt as your research requires change, with cost-effectiveness and highly researched report that larger companies can't match.

Information Safety
Market Research Update ensure that we deliver best reports. We care about the confidential and personal information quality, safety, of reports. We use Authorize secure payment process.

We Are Committed to Quality and Deadlines
We offer quality of reports within deadlines. We've worked hard to find the best ways to offer our customers results-oriented and process driven consulting services.

Our Remarkable Track Record
We concentrate on developing lasting and strong client relationship. At present, we hold numerous preferred relationships with industry leading firms that have relied on us constantly for their research requirements.

Best Service Assured
Buy reports from our executives that best suits your need and helps you stay ahead of the competition.

Customized Research Reports
Our research services are custom-made especially to you and your firm in order to discover practical growth recommendations and strategies. We don't stick to a one size fits all strategy. We appreciate that your business has particular research necessities.

Service Assurance
At Market Research Update, we are dedicated to offer the best probable recommendations and service to all our clients. You will be able to speak to experienced analyst who will be aware of your research requirements precisely.
Contact With Our Sales Team
Customer Testimonials
The content of the report is always up to the mark. Good to see speakers from expertise authorities.
Privacy requested , Managing Director
A lot of unique and interesting topics which are described in good manner.
Privacy requested, President
Well researched, expertise analysts, well organized, concrete and current topics delivered in time.
Privacy requested, Development Manager