Laboratory Automation Workcells Market Size, By Region (North America, Europe, Asia-Pacific, Latin America, Middle East and Africa), By Statistics, Trends, Outlook and Forecast 2026 to 2033 (Financial Impact Analysis)
ID : MRU_ 443821 | Date : Feb, 2026 | Pages : 243 | Region : Global | Publisher : MRU
Laboratory Automation Workcells Market Size
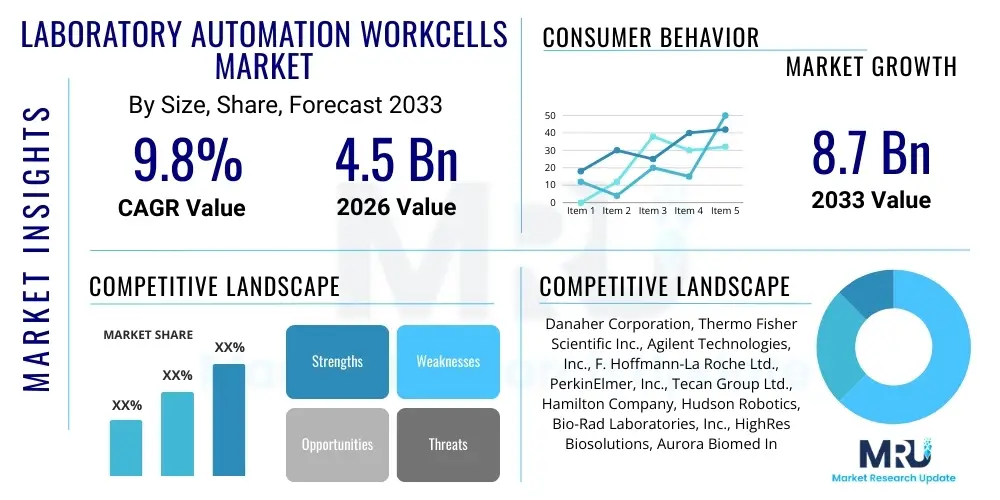
The Laboratory Automation Workcells Market is projected to grow at a Compound Annual Growth Rate (CAGR) of 9.8% between 2026 and 2033. The market is estimated at USD 4.5 Billion in 2026 and is projected to reach USD 8.7 Billion by the end of the forecast period in 2033.
Laboratory Automation Workcells Market introduction
The Laboratory Automation Workcells Market encompasses integrated systems designed to automate multiple complex laboratory processes, ranging from sample preparation and processing to analysis and data management. These workcells typically combine various robotic instruments, such as liquid handlers, plate readers, incubators, and robotic arms, coordinated by sophisticated scheduling software to execute high-throughput assays with minimal human intervention. This integration allows research and clinical laboratories to significantly increase efficiency, improve data quality, and accelerate discovery timelines, particularly in demanding fields like drug screening, genomics, and clinical diagnostics.
Laboratory automation workcells are instrumental in transitioning traditional manual lab operations into scalable, reliable, and standardized workflows. The primary applications span pharmaceutical research, biotechnology development, academic research institutions, and clinical microbiology labs. The inherent flexibility of modular workcells allows them to be reconfigured for different assay protocols, providing significant value in environments requiring rapid protocol adaptation and validation. These systems reduce the variability associated with human handling, ensuring consistency across large batches of samples, a critical requirement for regulatory compliance and robust scientific outcomes.
Key benefits derived from adopting these advanced systems include enhanced throughput capabilities, reduced operational costs in the long term, minimization of errors, and improved safety by limiting exposure to hazardous materials. The market is primarily driven by the escalating demand for high-throughput screening in drug discovery, the rapid advancements in genomics and personalized medicine requiring extensive sample processing, and the persistent need for efficiency improvements across the healthcare research ecosystem. Standardization and repeatability are core advantages that position automation workcells as indispensable tools for modern scientific advancement.
Laboratory Automation Workcells Market Executive Summary
The global Laboratory Automation Workcells Market is experiencing robust expansion, fundamentally driven by the pharmaceutical and biotech sectors' relentless pursuit of accelerated drug discovery pipelines and the burgeoning requirement for personalized medicine diagnostics. Current business trends indicate a strong shift towards modular and flexible workcell designs, which allow laboratories to scale their automation capabilities incrementally and adapt to evolving research needs. Furthermore, there is a pronounced focus on integrating advanced robotics and sophisticated control software that offer intuitive user interfaces and seamless connectivity with Laboratory Information Management Systems (LIMS), enhancing overall operational efficiency and data integrity across diverse lab environments.
Geographically, North America continues to dominate the market, propelled by substantial R&D investments, the presence of major pharmaceutical corporations, and widespread adoption of cutting-edge technologies. However, the Asia Pacific region is emerging as the fastest-growing market segment, primarily due to increasing government funding for life science research, rapid development of regional biotechnology firms, and growing demand for high-quality clinical diagnostics, especially in populous nations like China and India. European countries maintain a steady growth trajectory, supported by stringent regulatory frameworks that favor standardized, automated processes and collaborative research efforts funded by organizations like the European Union.
Segment trends reveal that integrated workcells, while having higher initial costs, are favored in high-throughput screening applications due to their optimized, end-to-end workflow capabilities. Conversely, modular workcells are gaining traction among smaller laboratories and academic institutions seeking cost-effective and flexible automation solutions. By application, drug discovery and clinical diagnostics remain the highest revenue generators, necessitating large-scale, automated platforms for compound testing, genetic sequencing, and routine clinical assay processing. End-user demand is consistently high from pharmaceutical and biotechnology companies, which leverage these systems to reduce time-to-market for new therapies.
AI Impact Analysis on Laboratory Automation Workcells Market
Common user questions regarding AI's influence on Laboratory Automation Workcells often center on how AI can enhance decision-making within automated workflows, optimize scheduling efficiency, and interpret the massive datasets generated by high-throughput instruments. Users frequently inquire about the feasibility of integrating AI for predictive maintenance, anticipating equipment failure before it occurs, and using machine learning algorithms to autonomously adjust experimental parameters in real-time to achieve optimal outcomes. There is also significant concern regarding data management and security, specifically how AI platforms handle sensitive research data and maintain compliance while driving operational intelligence. The overarching expectation is that AI will transform workcells from mere automated execution platforms into intelligent, self-optimizing discovery engines capable of accelerating scientific breakthroughs and significantly improving experimental reproducibility.
The primary impact of Artificial Intelligence (AI) integration is the shift from pre-programmed automation to intelligent automation. AI algorithms, particularly machine learning (ML), are being embedded into workcell scheduling software to dynamically manage queues, optimize robotic movements, and minimize idle time based on real-time data flow and resource availability. This level of optimization drastically improves throughput and reduces the operational bottlenecks often encountered in complex, multi-instrument workflows. Furthermore, AI facilitates automated quality control, identifying anomalies in plate images, liquid level sensing, or assay results, prompting immediate corrective action or flagging suspicious data points for human review, thereby ensuring higher data reliability.
Beyond operational enhancements, AI is revolutionizing the data analysis phase downstream of the workcell. High-throughput screening generates petabytes of complex biological and chemical data. AI platforms are essential for rapidly analyzing these voluminous datasets, identifying subtle patterns, classifying novel compounds, and predicting drug efficacy or toxicity with greater accuracy than traditional statistical methods. This predictive capacity accelerates lead optimization and candidate selection, making the automated workcell a truly indispensable component of the R&D cycle. The seamless integration of AI-powered data interpretation tools with the physical automation infrastructure is a key competitive differentiator in the modern laboratory setting.
- AI-driven optimization of robotic path planning and assay scheduling.
- Machine learning integration for predictive maintenance, minimizing unplanned downtime.
- Autonomous error detection and real-time parameter adjustment within workflows.
- Enhanced data analysis capabilities, including pattern recognition in high-content screening.
- Acceleration of hit-to-lead processes through AI-powered compound classification and prioritization.
- Development of self-calibrating and self-validating automation protocols.
DRO & Impact Forces Of Laboratory Automation Workcells Market
The Laboratory Automation Workcells Market is primarily propelled by the necessity for high-throughput screening (HTS) in pharmaceutical R&D, coupled with increasing investments in genomic research, demanding rapid and precise processing of vast sample volumes. However, significant restraints include the high initial capital expenditure required for purchasing and installing these complex integrated systems, alongside the technical challenge of ensuring seamless interoperability between different vendors' instruments within a single workcell. Opportunities arise from the ongoing shift towards personalized medicine, which necessitates flexible, automated diagnostic platforms, and the expansion into emerging markets, particularly in Asia, where research infrastructure is rapidly modernizing. These forces collectively shape the competitive landscape, pushing vendors toward modular solutions while simultaneously requiring robust technical support and training to maximize return on investment for end-users.
The primary driver is the intense competitive pressure in drug development, where pharmaceutical companies must accelerate candidate identification and validation. Laboratory automation workcells provide the essential infrastructure to execute millions of assays efficiently and reliably, directly impacting the speed and cost-effectiveness of preclinical development. This driver is amplified by global health crises and the subsequent push for rapid vaccine and therapeutic development, making scalable automation a critical priority. Furthermore, government initiatives worldwide supporting advanced life science research and precision medicine further fuel the demand for sophisticated, automated platforms capable of handling complex omics data generation.
Conversely, the high cost of implementation, encompassing specialized robotics, complex software licenses, and necessary infrastructure modifications, acts as a significant restraint, especially for smaller biotech startups and academic labs with constrained budgets. The complexity of integrating systems from multiple original equipment manufacturers (OEMs) often leads to extensive validation periods and specialized IT requirements, demanding highly skilled personnel for operation and maintenance. Nevertheless, the growing trend toward miniaturization of assays and the development of more affordable, flexible benchtop automation modules present clear opportunities, lowering the barrier to entry for smaller labs and expanding the potential customer base beyond major pharma corporations.
Segmentation Analysis
The Laboratory Automation Workcells Market is comprehensively segmented based on product type, application, and end-user, reflecting the diverse needs across the life science and clinical sectors. Product segmentation distinguishes between integrated workcells, which offer fully optimized, high-volume end-to-end solutions, and modular workcells, which provide flexibility for custom configurations and iterative expansion. This distinction is crucial as it dictates the scalability, initial investment, and complexity of deployment for the customer. Understanding these segments helps market players tailor their offerings, focusing on either high-throughput efficiency or adaptable modularity, depending on the targeted industry application.
Application segmentation reveals the core areas where automation delivers the most significant value, with drug discovery and development consistently dominating the market due to the sheer volume of assays required during screening and optimization phases. Genomic applications, including sequencing preparation and nucleic acid purification, represent a rapidly growing segment, driven by advances in gene therapy and personalized medicine. Furthermore, clinical diagnostics leverage automation workcells for routine tasks like sample accessioning, immunodiagnostics, and microbiology testing, ensuring regulatory compliance and maximizing patient sample throughput in hospital laboratories.
End-user analysis highlights pharmaceutical and biotechnology companies as the largest consumers, utilizing automation to accelerate time-to-market for novel therapeutics. Academic and research institutes also constitute a substantial segment, though often preferring modular, lower-cost solutions tailored for basic research and grant-funded projects. Clinical laboratories and hospitals represent the third key segment, prioritizing high reliability, regulatory traceability, and throughput for high-volume routine diagnostic testing. The structure of these segments underscores the market's dependence on sustained R&D expenditure and the ongoing modernization of global healthcare infrastructure.
- Product Type
- Integrated Workcells
- Modular Workcells
- Application
- Drug Discovery and Development
- Clinical Diagnostics
- Genomics and Proteomics
- High-Throughput Screening (HTS)
- Biobanking and Sample Management
- End-User
- Pharmaceutical and Biotechnology Companies
- Academic and Research Institutes
- Clinical Laboratories and Hospitals
- Components
- Robotic Systems (Robotic Arms, Track Systems)
- Software (Scheduling, LIMS Integration)
- Instrumentation (Liquid Handlers, Plate Readers, Incubators)
- Consumables and Reagents
Value Chain Analysis For Laboratory Automation Workcells Market
The value chain for the Laboratory Automation Workcells Market begins with the upstream suppliers responsible for providing highly specialized components, including precision robotics, optical sensors, microplate instrumentation, and high-purity chemical reagents. Key upstream activities involve advanced manufacturing and sourcing of mechanical and electronic parts, where quality control and technological innovation, particularly in miniaturization and speed, are paramount. Strategic partnerships between instrumentation manufacturers and specialized component providers are critical to ensuring the performance and reliability of the final integrated workcell system. The efficiency of this stage directly influences the cost and complexity of the final product, demanding robust supply chain management, especially given global sourcing challenges for sophisticated electronics.
The central stage of the value chain involves the Original Equipment Manufacturers (OEMs) and system integrators who design, assemble, configure, and validate the complex automation workcells. System integration is the most value-adding process, requiring expertise in software scheduling, robotic programming, and biological workflow understanding to create seamless, automated solutions. Distribution channels include direct sales forces, especially for highly customized, large-scale integrated systems sold to major pharmaceutical companies, and indirect channels such as specialized distributors and third-party resellers who cater to smaller labs and regional markets. The preference for direct engagement allows OEMs to offer comprehensive pre-sales consultation and post-installation support.
Downstream activities focus heavily on installation, validation, and continuous post-sales services, including maintenance, troubleshooting, and software updates. End-users—pharmaceutical companies, academic institutions, and clinical labs—are the final consumers, utilizing the workcells for specific high-throughput applications. Direct distribution ensures better control over customer relationships and service quality, which is vital for maintaining uptime in critical research and diagnostic environments. Indirect channels provide broader geographical reach, particularly in developing regions, but require rigorous training of reseller partners to maintain service standards. The success of the workcell relies heavily on robust service agreements and application support, ensuring that the system operates at peak efficiency throughout its lifecycle and that the investment yields the expected return.
Laboratory Automation Workcells Market Potential Customers
The primary end-users and buyers of laboratory automation workcells are institutions and corporations operating at the forefront of biological research, diagnostics, and pharmaceutical development who require large-scale, repeatable, and high-precision processing capabilities. Pharmaceutical and biotechnology firms represent the most lucrative customer segment, driven by the necessity to screen hundreds of thousands of potential drug candidates rapidly and manage clinical trial samples efficiently. These customers typically demand highly integrated, customized workcells capable of executing complex, multi-step assays, such as high-content imaging and compound management, often purchasing multiple units to support global R&D operations. Their buying decisions are heavily influenced by proven reliability, throughput metrics, and the vendor's capacity for deep software integration with enterprise systems like LIMS and ELN (Electronic Lab Notebooks).
Academic and government research institutes form a second, significant customer base. While their budgets are often tighter than corporate counterparts, requiring a preference for modular and flexible automation solutions, their demand is constant due to ongoing grant-funded projects in genomics, proteomics, and basic disease modeling. These institutions seek versatility, enabling them to reconfigure the workcell for different research protocols with minimal downtime. The purchasing criteria here often prioritize open-source compatibility, ease of programming, and robust technical support tailored to the educational environment. The transition from manual to automated processes in academic labs is accelerating due to the complexity of modern biological experiments.
Clinical diagnostic laboratories and hospital systems constitute the third major group of potential customers. Their requirements center on regulatory compliance (e.g., FDA, CLIA), maximum operational uptime, and high throughput for routine testing, such as blood chemistry, microbiology culturing, and molecular diagnostics (PCR testing). For clinical settings, automation workcells must ensure end-to-end sample traceability and rapid turnaround times critical for patient care. The key purchasing drivers include system validation, minimal maintenance complexity, and seamless integration with hospital information systems (HIS), ensuring patient data accuracy and rapid results delivery.
| Report Attributes | Report Details |
|---|---|
| Market Size in 2026 | USD 4.5 Billion |
| Market Forecast in 2033 | USD 8.7 Billion |
| Growth Rate | 9.8% CAGR |
| Historical Year | 2019 to 2024 |
| Base Year | 2025 |
| Forecast Year | 2026 - 2033 |
| DRO & Impact Forces |
|
| Segments Covered |
|
| Key Companies Covered | Danaher Corporation, Thermo Fisher Scientific Inc., Agilent Technologies, Inc., F. Hoffmann-La Roche Ltd., PerkinElmer, Inc., Tecan Group Ltd., Hamilton Company, Hudson Robotics, Bio-Rad Laboratories, Inc., HighRes Biosolutions, Aurora Biomed Inc., Siemens Healthineers, Abbot Laboratories, Brooks Automation, Inc., Cerner Corporation, QIAGEN N.V., SOTAX AG, Becton, Dickinson and Company. |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Enquiry Before Buy | Have specific requirements? Send us your enquiry before purchase to get customized research options. Request For Enquiry Before Buy |
Laboratory Automation Workcells Market Key Technology Landscape
The technological landscape of the Laboratory Automation Workcells Market is characterized by the convergence of advanced robotics, sophisticated liquid handling systems, and highly interconnected software platforms. Robotic systems, particularly articulated and collaborative robotic arms, are central to the workcell, providing precision movement, plate manipulation, and seamless transfer between instruments. The latest generation of robotics emphasizes speed, dexterity, and, increasingly, collaborative capabilities (cobots) that allow human operators to safely interact with the automated system for loading or maintenance without requiring extensive safety barriers. This evolution minimizes the footprint and enhances the operational flexibility of the workcell, making automation accessible for smaller laboratory spaces.
Liquid handling technology represents another critical pillar, utilizing automated pipetting systems that range from high-volume parallel dispensing to ultra-low volume nanoliter dispensing required for miniaturized assays. Advancements in non-contact dispensing technologies, such as acoustic dispensing, are driving efficiency by eliminating consumable waste and reducing contamination risks, which is vital in genomics and drug discovery. Furthermore, integrated sensor technologies, including optical sensors for liquid level detection and temperature/humidity control sensors, ensure the integrity of the experiment environment and provide real-time feedback for error handling and system monitoring. These technological refinements ensure high repeatability and accuracy, which are paramount for regulatory submissions.
Software and connectivity technologies are perhaps the most vital component in the modern workcell, enabling true automation intelligence. Key software includes high-level scheduling and control software that coordinates the actions of disparate instruments, ensuring timely execution and resource allocation. Crucially, seamless integration with Laboratory Information Management Systems (LIMS) and Electronic Lab Notebooks (ELN) is mandatory for data traceability, regulatory compliance, and efficient data exchange. The adoption of standardized communication protocols (such as SiLA or AnIML) facilitates better interoperability between different hardware and software modules, reducing the complexity of system customization and validation. This software infrastructure is foundational for future AI integration and remote monitoring capabilities.
Regional Highlights
The global Laboratory Automation Workcells Market demonstrates distinct regional dynamics, driven by varying levels of R&D investment, healthcare infrastructure maturity, and regulatory environments. North America, encompassing the United States and Canada, currently holds the largest market share. This dominance is attributed to the presence of global pharmaceutical and biotechnology giants, high expenditure on life science research, and early adoption of cutting-edge automation technologies, particularly in the fields of genomics and personalized medicine. The robust venture capital funding directed towards biotech startups in regions like Boston and San Francisco ensures sustained demand for high-end, customized automation solutions, driving market innovation and penetration across both corporate and academic settings.
Europe represents the second-largest market, characterized by mature healthcare systems and significant governmental support for advanced medical research, notably from organizations like the European Commission. Countries such as Germany, the UK, and Switzerland are major contributors, exhibiting strong demand from both clinical diagnostics labs focused on standardization and pharmaceutical companies adhering to stringent EU regulatory standards. The European market tends to favor reliability and validated performance, often driven by collaborative research projects requiring standardized, automated workflows. Growth is steady, focused on optimizing existing infrastructure and integrating modular solutions to enhance operational efficiency across large, publicly funded research institutions.
The Asia Pacific (APAC) region is forecasted to be the fastest-growing market segment throughout the forecast period. This rapid growth is fueled by increasing foreign direct investment in healthcare infrastructure, the expansion of local pharmaceutical manufacturing and R&D activities, and rising awareness regarding the benefits of laboratory efficiency in countries like China, India, Japan, and South Korea. Government initiatives aimed at modernizing clinical laboratories and promoting genomic research are key catalysts. While initial investment costs remain a hurdle, the increasing competitive pressure on regional pharmaceutical manufacturers to meet global quality standards is driving substantial adoption of cost-effective, high-throughput modular workcells, significantly boosting market growth potential.
- North America: Market leader due to large R&D budgets, high concentration of major pharma/biotech companies, and advanced clinical laboratory infrastructure.
- Europe: Stable growth driven by standardized diagnostics, strong academic research funding, and stringent quality regulations favoring automation consistency.
- Asia Pacific (APAC): Highest growth potential fueled by massive government investments in healthcare, expanding local biotech sector, and rapid modernization of clinical diagnostic capabilities.
- Latin America (LATAM): Emerging market showing gradual adoption, primarily focused on improving clinical diagnostic efficiency in major economies like Brazil and Mexico.
- Middle East and Africa (MEA): Limited but growing adoption, concentrated in Gulf Cooperation Council (GCC) countries investing heavily in advanced biomedical research infrastructure and specialized hospital facilities.
Top Key Players
The market research report includes a detailed profile of leading stakeholders in the Laboratory Automation Workcells Market.- Danaher Corporation
- Thermo Fisher Scientific Inc.
- Agilent Technologies, Inc.
- F. Hoffmann-La Roche Ltd.
- PerkinElmer, Inc.
- Tecan Group Ltd.
- Hamilton Company
- Hudson Robotics
- Bio-Rad Laboratories, Inc.
- HighRes Biosolutions
- Aurora Biomed Inc.
- Siemens Healthineers
- Abbot Laboratories
- Brooks Automation, Inc.
- Cerner Corporation
- QIAGEN N.V.
- SOTAX AG
- Becton, Dickinson and Company
- LGC Group
- Integra LifeSciences
Frequently Asked Questions
Analyze common user questions about the Laboratory Automation Workcells market and generate a concise list of summarized FAQs reflecting key topics and concerns.What is the projected CAGR for the Laboratory Automation Workcells Market between 2026 and 2033?
The Laboratory Automation Workcells Market is projected to exhibit a Compound Annual Growth Rate (CAGR) of 9.8% during the forecast period from 2026 to 2033, driven by increasing high-throughput screening demands.
Which end-user segment is the largest consumer of laboratory automation workcells?
Pharmaceutical and biotechnology companies represent the largest end-user segment, utilizing workcells extensively for accelerated drug discovery, compound screening, and clinical trial sample management due to the high volume and precision required.
How does AI technology enhance the utility of laboratory automation workcells?
AI enhances utility by enabling real-time optimization of robotic scheduling, predictive maintenance to prevent downtime, and advanced data analysis for rapid pattern recognition and anomaly detection in high-throughput datasets, transitioning automation to intelligent operations.
What are the main restraints impacting market growth?
The primary restraints are the substantial initial capital investment required for complex integrated systems and the operational challenges associated with software integration, validation, and the necessity for specialized technical personnel.
Which geographical region is expected to show the fastest growth rate?
The Asia Pacific (APAC) region is projected to register the fastest growth rate, fueled by substantial government investment in modernizing healthcare infrastructure and the rapid expansion of the indigenous biotechnology and pharmaceutical sectors.
What is the difference between integrated and modular automation workcells?
Integrated workcells are custom-built, fixed systems optimized for high volume, end-to-end workflows (e.g., mass HTS), whereas modular workcells are flexible, scalable platforms allowing labs to incrementally add components and reconfigure setups for diverse research protocols.
What types of instruments are typically included in a laboratory automation workcell?
A typical workcell integrates various instruments, including robotic arms, automated liquid handling systems (pipettors/dispensers), plate readers (absorbance, fluorescence), incubators, centrifuges, and barcode readers, all coordinated by specialized scheduling software.
How do workcells contribute to personalized medicine research?
Workcells are critical for personalized medicine by automating high-throughput genomic and proteomic sample preparation and analysis, allowing for the rapid and precise processing of patient-specific biological samples required for molecular diagnostics and gene therapy development.
What role does software play in modern workcell automation?
Software is essential for orchestration, providing high-level control over all integrated instruments, executing dynamic scheduling, managing error recovery, and crucially, ensuring seamless bidirectional data exchange and traceability with LIMS and ELN systems for regulatory compliance.
Are consumables a significant revenue component in this market?
Yes, while capital equipment dominates initial sales, specialized consumables, including microplates, disposable tips for liquid handlers, and specific reagents, generate a recurring and significant revenue stream throughout the operational lifecycle of the workcell.
What major technological advancement is currently shaping the workcell market?
The shift towards collaborative robotics (cobots) and advanced acoustic liquid handling systems is currently shaping the market, enhancing both safety and precision while allowing workcells to operate more efficiently in shared laboratory environments.
How do clinical laboratories benefit specifically from automation workcells?
Clinical laboratories benefit by achieving high sample throughput, reducing turnaround times for patient results, minimizing human error through standardization, and ensuring robust traceability required for accreditation and compliance in diagnostic testing environments.
Is the integration of systems from different vendors a common challenge?
Yes, achieving seamless interoperability between instruments sourced from different Original Equipment Manufacturers (OEMs) remains a persistent challenge, often requiring extensive custom programming and validation by system integrators to function efficiently as a cohesive workcell.
What is the primary driving factor originating from the pharmaceutical industry?
The primary driving factor is the intense and escalating need for High-Throughput Screening (HTS) capability, which necessitates automated platforms to process millions of chemical compounds and biological samples quickly and reliably during the early stages of drug discovery.
How do regulatory requirements influence the adoption of automation workcells?
Regulatory bodies often favor automated processes because they ensure consistency, repeatability, and complete audit trails for every step of an experiment or diagnostic procedure, making validation and compliance easier compared to manual operations, thus accelerating adoption.
What are the key concerns regarding data security in automated labs?
Key data security concerns revolve around protecting sensitive research IP and patient data stored within LIMS/ELN systems connected to the workcells, particularly with increased connectivity and remote monitoring capabilities that broaden the potential attack surface.
How does the market address the need for flexible automation in academic settings?
The market addresses this by developing highly modular and easily reconfigurable workcell systems. These flexible solutions allow academic labs to reuse components across different research projects and align automation capabilities with variable grant funding cycles.
What impact does miniaturization of assays have on workcell design?
Miniaturization drives the need for ultra-precise nanoliter liquid handling systems and high-resolution imaging capabilities within the workcell, demanding more compact and sensitive instruments to accurately handle and analyze reduced sample volumes.
What is the role of the Value Chain's upstream segment in the market?
The upstream segment involves suppliers of specialized, high-precision components like robotics, sensors, and electronic controls. Their innovation in speed, precision, and component quality directly dictates the performance and cost of the final integrated workcell system.
How is the market leveraging the Internet of Things (IoT)?
The market leverages IoT by implementing network-connected sensors and instruments across the workcell to enable real-time monitoring, remote diagnostics, predictive maintenance scheduling, and centralized data logging across multiple geographically dispersed laboratory facilities.
Which segment currently accounts for the largest market share by product type?
While both segments are strong, integrated workcells typically account for the largest revenue share, primarily due to their high cost, comprehensive feature set, and mandatory use in large-scale, dedicated high-throughput screening operations by major pharmaceutical entities.
What types of training are required for operating laboratory workcells?
Operators require specialized training covering robotic programming, software scheduling, troubleshooting instrument malfunctions, assay protocol validation, and adherence to specific safety and quality assurance protocols inherent to automated high-throughput systems.
Is biobanking a major application for workcells?
Yes, biobanking is a crucial application. Workcells automate the processing, storage, retrieval, and tracking of large collections of biological samples, ensuring high sample integrity, accurate inventory management, and reliable long-term sample viability.
How is environmental sustainability addressed by workcell manufacturers?
Manufacturers are addressing sustainability by designing systems that utilize less power, minimize the use of consumables and reagents through miniaturized assays, and incorporate efficient waste management systems, aligning with corporate social responsibility goals.
What is the expected long-term impact of workcell adoption on operational costs?
While initial capital costs are high, the long-term impact on operational costs is favorable, characterized by significant reductions in labor costs, decreased consumption of costly reagents (due to miniaturization), and fewer errors requiring re-testing, leading to a strong return on investment.
How does the presence of leading pharma companies influence North America's market dominance?
The high concentration of global pharmaceutical headquarters in North America drives massive investment into advanced R&D infrastructure, leading to consistent demand for the latest, most sophisticated, and often customized, high-cost laboratory automation workcells, thus securing regional dominance.
What is a key differentiator in the competitive landscape of automation workcells?
A key differentiator is the proprietary software platform's ability to seamlessly integrate diverse third-party hardware components and provide intuitive, flexible scheduling and data analysis capabilities (interoperability and software intelligence).
How do workcells aid in microbiology and infectious disease diagnostics?
In microbiology, workcells automate sample preparation, culture incubation, colony picking, and high-throughput molecular testing (PCR), significantly speeding up the identification of pathogens and improving response times during infectious disease outbreaks.
What role does standardization play in the market?
Standardization is crucial for market expansion, particularly through initiatives like SiLA (Standardization in Lab Automation), which promote common communication protocols, making it easier for labs to integrate instruments from multiple vendors and simplify system development and validation.
How are emerging markets like India and China impacting the market?
Emerging markets are primarily driving the demand for modular, cost-effective automation solutions as their pharmaceutical and clinical sectors rapidly modernize, increasing the overall volume and global accessibility of entry-level and mid-range workcell systems.
To check our Table of Contents, please mail us at: sales@marketresearchupdate.com
Research Methodology
The Market Research Update offers technology-driven solutions and its full integration in the research process to be skilled at every step. We use diverse assets to produce the best results for our clients. The success of a research project is completely reliant on the research process adopted by the company. Market Research Update assists its clients to recognize opportunities by examining the global market and offering economic insights. We are proud of our extensive coverage that encompasses the understanding of numerous major industry domains.
Market Research Update provide consistency in our research report, also we provide on the part of the analysis of forecast across a gamut of coverage geographies and coverage. The research teams carry out primary and secondary research to implement and design the data collection procedure. The research team then analyzes data about the latest trends and major issues in reference to each industry and country. This helps to determine the anticipated market-related procedures in the future. The company offers technology-driven solutions and its full incorporation in the research method to be skilled at each step.
The Company's Research Process Has the Following Advantages:
- Information Procurement
The step comprises the procurement of market-related information or data via different methodologies & sources.
- Information Investigation
This step comprises the mapping and investigation of all the information procured from the earlier step. It also includes the analysis of data differences observed across numerous data sources.
- Highly Authentic Source
We offer highly authentic information from numerous sources. To fulfills the client’s requirement.
- Market Formulation
This step entails the placement of data points at suitable market spaces in an effort to assume possible conclusions. Analyst viewpoint and subject matter specialist based examining the form of market sizing also plays an essential role in this step.
- Validation & Publishing of Information
Validation is a significant step in the procedure. Validation via an intricately designed procedure assists us to conclude data-points to be used for final calculations.
×
Request Free Sample:
Related Reports
Select License

Why Choose Us

We're cost-effective and Offered Best services:
We are flexible and responsive startup research firm. We adapt as your research requires change, with cost-effectiveness and highly researched report that larger companies can't match.

Information Safety
Market Research Update ensure that we deliver best reports. We care about the confidential and personal information quality, safety, of reports. We use Authorize secure payment process.

We Are Committed to Quality and Deadlines
We offer quality of reports within deadlines. We've worked hard to find the best ways to offer our customers results-oriented and process driven consulting services.

Our Remarkable Track Record
We concentrate on developing lasting and strong client relationship. At present, we hold numerous preferred relationships with industry leading firms that have relied on us constantly for their research requirements.

Best Service Assured
Buy reports from our executives that best suits your need and helps you stay ahead of the competition.

Customized Research Reports
Our research services are custom-made especially to you and your firm in order to discover practical growth recommendations and strategies. We don't stick to a one size fits all strategy. We appreciate that your business has particular research necessities.

Service Assurance
At Market Research Update, we are dedicated to offer the best probable recommendations and service to all our clients. You will be able to speak to experienced analyst who will be aware of your research requirements precisely.
Contact With Our Sales Team
Customer Testimonials
The content of the report is always up to the mark. Good to see speakers from expertise authorities.
Privacy requested , Managing Director
A lot of unique and interesting topics which are described in good manner.
Privacy requested, President
Well researched, expertise analysts, well organized, concrete and current topics delivered in time.
Privacy requested, Development Manager
