Levothyroxine Sodium Tablet Market Size, By Region (North America, Europe, Asia-Pacific, Latin America, Middle East and Africa), By Statistics, Trends, Outlook and Forecast 2026 to 2033 (Financial Impact Analysis)
ID : MRU_ 443425 | Date : Feb, 2026 | Pages : 251 | Region : Global | Publisher : MRU
Levothyroxine Sodium Tablet Market Size
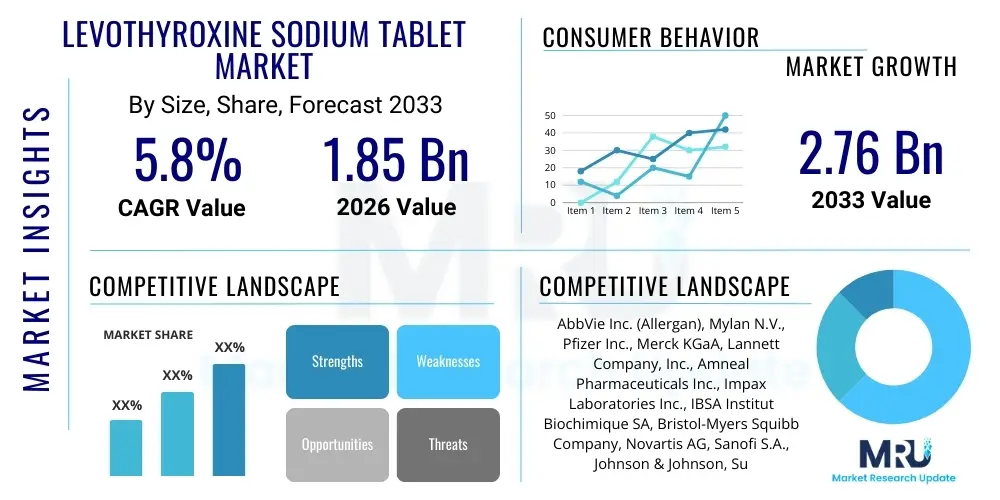
The Levothyroxine Sodium Tablet Market is projected to grow at a Compound Annual Growth Rate (CAGR) of 5.8% between 2026 and 2033. The market is estimated at USD 1.85 Billion in 2026 and is projected to reach USD 2.76 Billion by the end of the forecast period in 2033.
Levothyroxine Sodium Tablet Market introduction
The Levothyroxine Sodium Tablet market encompasses prescription pharmaceuticals used primarily for the treatment of hypothyroidism, a condition resulting from insufficient production of thyroid hormones by the thyroid gland. Levothyroxine sodium is a synthetic version of the naturally occurring thyroid hormone, T4 (thyroxine), which is essential for regulating metabolism, energy levels, and overall bodily functions. These tablets are critical for maintaining euthyroid status in patients across all age groups and demographics who suffer from chronic thyroid insufficiency. The global adoption is driven significantly by the rising prevalence of autoimmune diseases like Hashimoto’s thyroiditis, increased geriatric populations susceptible to thyroid disorders, and enhanced diagnostic capabilities leading to earlier detection of subclinical hypothyroidism.
The primary applications of levothyroxine sodium tablets include treatment for primary, secondary, and tertiary congenital or acquired hypothyroidism, management of thyroid stimulating hormone (TSH) suppression in patients with thyroid cancer, and as an adjunct to surgery and radioiodine therapy for certain thyroid conditions. Due to its narrow therapeutic index, achieving and maintaining the correct dosage requires diligent monitoring and individualized treatment plans. The key benefit of this product lies in its ability to effectively replace the deficient endogenous hormone, thereby resolving debilitating symptoms such as fatigue, weight gain, depression, and cognitive impairment, significantly improving the patient's quality of life and preventing serious long-term cardiovascular and metabolic complications associated with untreated thyroid dysfunction.
Major driving factors influencing the market trajectory include robust governmental health programs promoting chronic disease management, consistent innovation in stable tablet formulations to improve bioavailability, and the expansion of healthcare infrastructure in emerging economies. Furthermore, growing public health awareness regarding thyroid health, coupled with routine screening protocols, particularly in developed regions like North America and Europe, continues to bolster prescription rates. While patent expiries have introduced strong generic competition, the reliance on established brand names in some high-income regions, driven by physician and patient trust in consistent formulation and efficacy, sustains significant market value, contributing to steady revenue streams throughout the forecast period.
Levothyroxine Sodium Tablet Market Executive Summary
The global Levothyroxine Sodium Tablet market is characterized by stability driven by chronic disease prevalence, yet intensely competitive due to the dominance of generic formulations. Business trends indicate a focus on manufacturing excellence, quality control, and supply chain optimization, crucial elements given the product's narrow therapeutic window and the necessity for precise dosing. Key players are navigating the balance between maintaining strong brand recognition for originator products (such as Synthroid and Euthyrox) and managing price erosion from high-volume generic equivalents. Strategic partnerships aiming at localized manufacturing and distribution are becoming prevalent, especially to penetrate rapidly growing markets in Asia Pacific and Latin America, addressing localized regulatory requirements and expanding patient access.
Regionally, North America maintains the largest market share, attributed to high diagnostic rates, established reimbursement frameworks, and a historically high consumption volume of branded and authorized generic versions. However, the Asia Pacific region is anticipated to demonstrate the fastest growth rate, fueled by improving healthcare access, increasing screening campaigns, and the sheer volume of its burgeoning population base, many of whom are entering age brackets prone to thyroid disorders. European markets show maturity, focusing on standardized treatment guidelines and cost-containment measures, heavily favoring bioequivalent generic substitutions where possible, though specialized formulations for specific patient demographics (e.g., pediatric or elderly patients) retain niche value.
Segmentation trends highlight the continued dominance of the standard 25 mcg to 150 mcg dose ranges, reflecting the most commonly prescribed maintenance doses. Within formulation types, oral tablets remain the predominant delivery method, though attention is being paid to specialized liquid formulations for patients facing swallowing difficulties or requiring highly flexible dose adjustments, though these currently constitute a small fraction of the total market. From an end-user perspective, retail pharmacies and hospital pharmacies are the primary distribution channels, with retail channels capturing the majority due to the chronic, long-term nature of treatment. The interplay between regulatory bodies promoting generic use and patient adherence to established treatment protocols defines the financial landscape of this highly necessary pharmaceutical segment.
AI Impact Analysis on Levothyroxine Sodium Tablet Market
User inquiries regarding the impact of Artificial Intelligence (AI) on the Levothyroxine Sodium Tablet market predominantly revolve around personalized dosing strategies, optimization of drug discovery and manufacturing processes, and improvements in patient monitoring for adherence and adverse effects. Key concerns often address whether AI can solve the long-standing challenge of dosage variability in levothyroxine treatment, which is critical due to the drug's narrow therapeutic index. Users are particularly interested in AI-driven diagnostic tools that can predict patient response based on genetic and metabolic profiles, moving beyond standard TSH monitoring. Expectations center on AI enhancing efficiency, reducing medication errors, and potentially speeding up the development of more stable or novel synthetic thyroid hormone alternatives, while also addressing supply chain vulnerabilities through predictive logistics.
The application of machine learning algorithms in clinical settings is poised to revolutionize how levothyroxine treatment is initiated and maintained. AI systems can process massive datasets, including patient demographics, co-morbidities, drug interactions, and real-time TSH measurements, to recommend highly personalized initial doses and subsequent titration adjustments, significantly reducing the time required for patients to reach euthyroid status. This capability addresses a major pain point for endocrinologists and general practitioners, enhancing treatment efficacy and patient satisfaction, subsequently reducing the healthcare burden associated with suboptimal thyroid control. This precision medicine approach, driven by AI, is anticipated to improve overall treatment adherence.
Furthermore, AI is making inroads into the operational aspects of the market, particularly in quality control and manufacturing. Computer vision systems and predictive modeling are being utilized to monitor tablet manufacturing lines, ensuring consistency in active pharmaceutical ingredient (API) dispersion, tablet weight, and dissolution profiles—crucial factors for a drug requiring high bioequivalence. In the supply chain, AI algorithms forecast demand spikes based on regional health data and seasonal fluctuations, optimizing inventory management and mitigating potential shortages, ensuring a steady supply of this essential, life-sustaining medication across global distribution networks, thereby bolstering market resilience and operational profitability for large generic manufacturers.
- AI optimizes personalized levothyroxine dosing based on genetic and clinical data, improving TSH normalization speed.
- Machine learning enhances diagnostic accuracy for subclinical and complex hypothyroidism cases.
- Predictive maintenance and quality control in manufacturing leverage AI to ensure high tablet consistency and bioequivalence.
- AI-driven supply chain logistics improve inventory forecasting, reducing stock-outs of critical medication.
- Natural Language Processing (NLP) assists in pharmacovigilance by analyzing patient feedback and adverse event reports efficiently.
DRO & Impact Forces Of Levothyroxine Sodium Tablet Market
The dynamics of the Levothyroxine Sodium Tablet market are governed by a robust interplay of driving forces, significant regulatory restraints, and ample opportunities arising from therapeutic expansion and geographic penetration. The primary driver is the pervasive, growing global prevalence of hypothyroidism, especially among aging populations and in regions experiencing lifestyle shifts that contribute to autoimmune conditions. This constant, non-negotiable demand for chronic, daily therapy ensures market stability and predictable growth. However, this stability is counterbalanced by intense pricing pressure stemming from the market maturity and the fierce competition among generic manufacturers, mandated by payors and governments prioritizing cost efficiency in chronic care. Opportunities exist in developing stable, non-standard formulations and improving patient adherence through digital health integration.
Key drivers include increased awareness campaigns leading to higher screening rates, successful integration of thyroid function tests into routine health check-ups, and the expansion of treatment guidelines globally that standardize the long-term management of subclinical hypothyroidism. Conversely, the market faces significant restraints, chiefly regulatory hurdles regarding bioequivalence studies for generic products. Because of the drug’s narrow therapeutic index, even minor variations between generic and branded products are scrutinized heavily, leading to long approval processes and patient/physician hesitancy toward switching formulations. Additionally, the challenge of patient non-adherence, although not unique to levothyroxine, remains a restraint on optimal utilization, often leading to increased healthcare costs related to uncontrolled thyroid conditions.
The impact forces shaping the market are multifaceted, encompassing macroeconomic stability, regulatory policy shifts, and technological advancements. Regulatory forces, particularly the stringent requirements set by agencies like the FDA and EMA for product quality and batch consistency, exert a powerful influence, dictating market entry and operational costs. Economic forces dictate the dominance of generics, with payor strategies emphasizing cost-effective chronic care, leading to constant price negotiations and downward pressure on Average Selling Prices (ASPs). Finally, technological forces related to advanced drug delivery systems (e.g., liquid gels or precision micro-dosing tablets) offer opportunities for premium pricing and differentiation, particularly appealing to patients who struggle with standard oral tablet routines or require highly stable formulations, allowing niche growth despite the overall generic landscape.
Segmentation Analysis
The Levothyroxine Sodium Tablet market is comprehensively segmented based on three critical parameters: dosage strength, which dictates therapeutic application and patient demographic; end-user, identifying the key consumption points; and distribution channel, which maps the logistical pathways from manufacturer to patient. Understanding these segments is paramount for strategic planning, as it reveals the concentration of value and volume across different market niches. The market structure reflects the essential, non-discretionary nature of the product, with segment growth largely tracking demographic changes and regional differences in healthcare accessibility and prescribing habits. High-volume generic consumption defines most segments, yet premium pricing is sustained in specific, high-compliance brand segments.
Segmentation by dosage strength is particularly instructive, showing a high concentration in mid-range strengths (50 mcg to 125 mcg) which represent standard maintenance doses for adult hypothyroid patients. Smaller doses (e.g., 25 mcg) cater primarily to initial titration phases or patients with subclinical conditions, while higher doses (above 150 mcg) are reserved for more severe cases, postsurgical TSH suppression, or large-statured individuals. The end-user segmentation clearly indicates that retail pharmacies hold the lion's share, given that hypothyroidism is a chronic condition managed predominantly on an outpatient basis. Conversely, hospital pharmacies manage the initial treatment phases, acute care settings, and potentially administer specialized intravenous forms (though not tablets) in emergent situations, maintaining a significant procurement role.
Analyzing the distribution channels reveals the intricate supply chain, which is heavily reliant on pharmaceutical wholesalers and distributors who bridge the gap between large-scale manufacturers and numerous retail outlets. Direct distribution models are rare, reserved mainly for specialized or limited-access markets. The robustness of the generic supply chain relies heavily on high-efficiency third-party logistics (3PL) providers capable of handling massive volumes at low margins. Strategic focus remains on ensuring supply chain integrity, minimizing cold chain risks (where applicable for specific storage conditions), and maintaining track-and-trace capabilities to comply with global regulatory requirements, ensuring patient safety and product authenticity in a highly fragmented distribution network, particularly in developing markets.
- By Dosage Strength:
- 25 mcg
- 50 mcg
- 75 mcg
- 88 mcg
- 100 mcg
- 112 mcg
- 125 mcg
- 150 mcg and Above
- By End-User:
- Retail Pharmacies
- Hospital Pharmacies
- Online Pharmacies
- By Distribution Channel:
- Wholesalers and Distributors
- Direct Sales
- Others (e.g., Governmental Procurement Agencies)
Value Chain Analysis For Levothyroxine Sodium Tablet Market
The value chain for Levothyroxine Sodium Tablets begins with the upstream segment, primarily involving the sourcing and synthesis of the Active Pharmaceutical Ingredient (API), Levothyroxine Sodium. This process requires highly specialized chemical synthesis capabilities and stringent quality control, as API stability and purity are critical determinants of the final product’s efficacy and bioequivalence. Manufacturers often rely on a limited number of specialized global API suppliers, particularly those based in Asia (e.g., India and China) who offer cost efficiencies, though branded manufacturers often maintain tighter control over their source materials to minimize batch variation. Key challenges in this stage include regulatory compliance for Good Manufacturing Practices (GMP) and ensuring environmental sustainability in chemical processing, which significantly impacts input costs and supply resilience across the industry.
Moving downstream, the value chain encompasses formulation, tablet manufacturing, quality assurance, packaging, and subsequent distribution. Formulation complexity involves optimizing excipients to ensure tablet stability and desired dissolution rates, crucial elements for absorption. Once manufactured, the products enter a highly regulated distribution channel. Direct distribution (manufacturer to large hospital systems or government programs) exists but is less common. The indirect channel, managed predominantly by pharmaceutical wholesalers and distributors, handles the majority of the volume. These entities manage inventory, logistics, and regional compliance, effectively bridging the gap between centralized manufacturing sites and highly fragmented endpoints such as retail pharmacies and smaller healthcare facilities. Effective supply chain management is key to minimizing stock-outs and ensuring timely access for patients.
The final stages involve prescribing and consumption. Physicians (endocrinologists, internal medicine specialists, and general practitioners) are the primary gatekeepers, initiating treatment and managing ongoing dosing. Retail pharmacies serve as the immediate point of sale and patient interaction, often providing crucial adherence counseling and managing prescription refills. Due to the chronic nature of hypothyroidism, repeat purchasing is guaranteed, lending high stability to the revenue stream. Payors (government health agencies, private insurance, and managed care organizations) exert strong influence throughout the chain, utilizing formularies and reimbursement policies to drive generic usage, thereby continuously applying pricing pressure on both branded and generic manufacturers, forcing optimization of cost structures across all stages of production and distribution.
Levothyroxine Sodium Tablet Market Potential Customers
The primary consumers and end-users of Levothyroxine Sodium Tablets are patients diagnosed with hypothyroidism, requiring chronic, life-long treatment. This vast patient pool spans all demographics, from infants diagnosed with congenital hypothyroidism to a significant proportion of the elderly population, who often develop subclinical or overt hypothyroidism due to aging or co-morbid autoimmune conditions. Healthcare providers, including endocrinologists, general practitioners, and primary care physicians, act as the crucial intermediaries who initiate and manage the therapeutic relationship, effectively driving the prescription volume. Therefore, manufacturers focus their marketing and educational outreach programs both toward these prescribing authorities and to the institutional purchasers.
Institutional buyers represent the major procurement entities in the market. This group includes large hospital systems, specialized endocrine clinics, governmental procurement agencies (especially in countries with national health services), and managed care organizations (MCOs) or Pharmacy Benefit Managers (PBMs) in highly insured markets like the United States. These organizations procure medication in bulk, and their purchasing decisions are heavily influenced by cost-effectiveness, contract pricing, and demonstrated bioequivalence data, driving intense competition among generic suppliers. The sheer volume aggregated by these buyers makes them critical targets for large-scale pharmaceutical sales operations, often dictating the market share distribution between competing formulations.
Finally, the growing segment of online pharmacies and telehealth services represents an expanding customer base, particularly for routine prescription refills. As chronic disease management increasingly leverages digital platforms, patients utilize online pharmacies for convenience and often lower out-of-pocket costs. These platforms require robust, efficient distribution logistics and compliant digital verification systems. Targeting these emerging channels allows manufacturers to optimize last-mile delivery and enhance patient engagement, especially in geographically dispersed or underserved areas, reflecting a future shift in retail drug purchasing behavior away from traditional brick-and-mortar pharmacies for chronic medication management.
| Report Attributes | Report Details |
|---|---|
| Market Size in 2026 | USD 1.85 Billion |
| Market Forecast in 2033 | USD 2.76 Billion |
| Growth Rate | 5.8% CAGR |
| Historical Year | 2019 to 2024 |
| Base Year | 2025 |
| Forecast Year | 2026 - 2033 |
| DRO & Impact Forces |
|
| Segments Covered |
|
| Key Companies Covered | AbbVie Inc. (Allergan), Mylan N.V., Pfizer Inc., Merck KGaA, Lannett Company, Inc., Amneal Pharmaceuticals Inc., Impax Laboratories Inc., IBSA Institut Biochimique SA, Bristol-Myers Squibb Company, Novartis AG, Sanofi S.A., Johnson & Johnson, Sun Pharmaceutical Industries Ltd., Teva Pharmaceutical Industries Ltd., Alvogen, Inc., Strides Pharma Science Limited, Sandoz (Novartis subsidiary), Aspen Pharmacare Holdings Limited, Cipla Ltd., Zydus Cadila. |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Enquiry Before Buy | Have specific requirements? Send us your enquiry before purchase to get customized research options. Request For Enquiry Before Buy |
Levothyroxine Sodium Tablet Market Key Technology Landscape
The technological landscape for the Levothyroxine Sodium Tablet market is primarily centered on enhancing manufacturing precision, ensuring long-term drug stability, and exploring alternative delivery methods to improve patient compliance and absorption consistency. A major area of focus involves advanced tablet compression techniques and pharmaceutical coating technologies designed to protect the highly sensitive levothyroxine API from degradation caused by moisture, light, and temperature fluctuations, which can alter the drug’s potency and shelf life. Manufacturers are investing in high-speed, automated manufacturing lines integrated with Process Analytical Technology (PAT) tools, allowing for real-time monitoring of critical quality attributes such as blend uniformity and tablet hardness, ensuring every batch meets the stringent bioequivalence standards necessary for this narrow therapeutic index drug.
Beyond traditional manufacturing, innovation is seen in the development of specialized formulations intended to overcome existing patient challenges. This includes the implementation of stable liquid levothyroxine formulations, which utilize advanced solubilization technology and specific preservative systems. These liquid options are particularly beneficial for pediatric populations, patients with swallowing difficulties (dysphagia), or those undergoing procedures that affect gastrointestinal absorption, offering a more flexible and potentially more rapidly absorbed dose. However, the manufacturing complexity and higher cost associated with maintaining stability in liquid forms compared to solid tablets currently limit their market penetration, making solid dosage forms technologically and economically dominant.
Furthermore, technology plays a critical role in the supporting infrastructure, including advanced packaging solutions utilizing blister packs with superior moisture barriers and light-protective materials (e.g., specialized aluminum foil). Digital technologies, such as smart blister packaging or associated mobile applications, are being explored to track patient adherence and provide reminders, linking the physical product to digital health ecosystems. While Levothyroxine itself is a legacy molecule, the continuous technological upgrades in its delivery, manufacturing quality control (CQM), and supply chain management are essential competitive differentiators, particularly as generic producers race to demonstrate superior and consistent quality to capture market share from established branded products.
Regional Highlights
The Levothyroxine Sodium Tablet market exhibits distinct regional dynamics heavily influenced by healthcare policies, disease prevalence, and demographic structures.
- North America (NA): Dominates the market due to the high incidence of thyroid disorders, comprehensive screening protocols, and sophisticated healthcare infrastructure. The US market, in particular, is significant, characterized by a mix of highly popular branded products (like Synthroid) and strong generic competition. High drug expenditure and established reimbursement mechanisms ensure consistent prescription volumes, although pricing pressures from PBMs are perpetually high.
- Europe: A mature market defined by rigorous regulatory standards (EMA) and a strong emphasis on cost-containment through national health services. Germany, the UK, and France are major contributors, with generic substitution rates being extremely high. The region focuses heavily on ensuring batch consistency and bioequivalence compliance due to historical scrutiny of generic thyroid medications.
- Asia Pacific (APAC): Expected to register the highest growth rate, driven by massive population density, increasing urbanization, rising awareness of chronic diseases, and improving access to diagnostic services, especially in China, India, and Southeast Asia. The market is fragmented, presenting significant opportunities for generic manufacturers to scale operations and benefit from government initiatives aimed at expanding access to essential medicines.
- Latin America (LATAM): Growth is steady, supported by improving economies and expanding health coverage, particularly in Brazil and Mexico. The market often favors imported formulations, though local production is rising. Political and economic instability can sometimes affect drug procurement and supply chain continuity, posing unique challenges compared to developed markets.
- Middle East and Africa (MEA): Represents a nascent market with diverse needs. Growth is concentrated in the Gulf Cooperation Council (GCC) countries due to high per capita healthcare spending and government investment in chronic disease management. African sub-regions face challenges related to infrastructure and access, though partnerships targeting large patient populations for essential medicines are growing.
Top Key Players
The market research report includes a detailed profile of leading stakeholders in the Levothyroxine Sodium Tablet Market.- AbbVie Inc. (Allergan)
- Mylan N.V.
- Pfizer Inc.
- Merck KGaA
- Lannett Company, Inc.
- Amneal Pharmaceuticals Inc.
- Impax Laboratories Inc.
- IBSA Institut Biochimique SA
- Bristol-Myers Squibb Company
- Novartis AG
- Sanofi S.A.
- Johnson & Johnson
- Sun Pharmaceutical Industries Ltd.
- Teva Pharmaceutical Industries Ltd.
- Alvogen, Inc.
- Strides Pharma Science Limited
- Sandoz (Novartis subsidiary)
- Aspen Pharmacare Holdings Limited
- Cipla Ltd.
- Zydus Cadila
Frequently Asked Questions
Analyze common user questions about the Levothyroxine Sodium Tablet market and generate a concise list of summarized FAQs reflecting key topics and concerns.What is the primary factor driving the growth of the Levothyroxine Sodium Tablet market?
The predominant driver is the increasing global prevalence of hypothyroidism, an irreversible and chronic condition requiring continuous, lifelong hormone replacement therapy, coupled with rising public and clinical awareness leading to higher diagnostic rates.
Why is bioequivalence a critical issue in the Levothyroxine Sodium Tablet market?
Levothyroxine has a narrow therapeutic index, meaning small changes in drug absorption or potency can lead to significant clinical consequences. Therefore, generic versions must demonstrate strict bioequivalence to ensure consistent and safe patient outcomes compared to branded formulations.
Which geographical region holds the largest market share for Levothyroxine Sodium Tablets?
North America currently holds the largest market share, driven by high prescribing rates, comprehensive insurance coverage, and a historically entrenched preference for established branded and authorized generic medications for chronic conditions.
How is technological innovation affecting this market?
Technological innovation focuses primarily on improving manufacturing precision (PAT systems) for tablet consistency, developing highly stable liquid formulations for specific patient groups (e.g., pediatrics), and utilizing AI for personalized, precise dosing recommendations.
What are the main restraints on market profitability despite high demand?
The primary restraint is intense price erosion caused by widespread generic competition and aggressive procurement strategies by pharmacy benefit managers (PBMs) and governmental payors aiming for cost containment in chronic prescription expenditures.
To check our Table of Contents, please mail us at: sales@marketresearchupdate.com
Research Methodology
The Market Research Update offers technology-driven solutions and its full integration in the research process to be skilled at every step. We use diverse assets to produce the best results for our clients. The success of a research project is completely reliant on the research process adopted by the company. Market Research Update assists its clients to recognize opportunities by examining the global market and offering economic insights. We are proud of our extensive coverage that encompasses the understanding of numerous major industry domains.
Market Research Update provide consistency in our research report, also we provide on the part of the analysis of forecast across a gamut of coverage geographies and coverage. The research teams carry out primary and secondary research to implement and design the data collection procedure. The research team then analyzes data about the latest trends and major issues in reference to each industry and country. This helps to determine the anticipated market-related procedures in the future. The company offers technology-driven solutions and its full incorporation in the research method to be skilled at each step.
The Company's Research Process Has the Following Advantages:
- Information Procurement
The step comprises the procurement of market-related information or data via different methodologies & sources.
- Information Investigation
This step comprises the mapping and investigation of all the information procured from the earlier step. It also includes the analysis of data differences observed across numerous data sources.
- Highly Authentic Source
We offer highly authentic information from numerous sources. To fulfills the client’s requirement.
- Market Formulation
This step entails the placement of data points at suitable market spaces in an effort to assume possible conclusions. Analyst viewpoint and subject matter specialist based examining the form of market sizing also plays an essential role in this step.
- Validation & Publishing of Information
Validation is a significant step in the procedure. Validation via an intricately designed procedure assists us to conclude data-points to be used for final calculations.
×
Request Free Sample:
Related Reports
Select License

Why Choose Us

We're cost-effective and Offered Best services:
We are flexible and responsive startup research firm. We adapt as your research requires change, with cost-effectiveness and highly researched report that larger companies can't match.

Information Safety
Market Research Update ensure that we deliver best reports. We care about the confidential and personal information quality, safety, of reports. We use Authorize secure payment process.

We Are Committed to Quality and Deadlines
We offer quality of reports within deadlines. We've worked hard to find the best ways to offer our customers results-oriented and process driven consulting services.

Our Remarkable Track Record
We concentrate on developing lasting and strong client relationship. At present, we hold numerous preferred relationships with industry leading firms that have relied on us constantly for their research requirements.

Best Service Assured
Buy reports from our executives that best suits your need and helps you stay ahead of the competition.

Customized Research Reports
Our research services are custom-made especially to you and your firm in order to discover practical growth recommendations and strategies. We don't stick to a one size fits all strategy. We appreciate that your business has particular research necessities.

Service Assurance
At Market Research Update, we are dedicated to offer the best probable recommendations and service to all our clients. You will be able to speak to experienced analyst who will be aware of your research requirements precisely.
Contact With Our Sales Team
Customer Testimonials
The content of the report is always up to the mark. Good to see speakers from expertise authorities.
Privacy requested , Managing Director
A lot of unique and interesting topics which are described in good manner.
Privacy requested, President
Well researched, expertise analysts, well organized, concrete and current topics delivered in time.
Privacy requested, Development Manager
