Medical Disposable Gloves Market Size, By Region (North America, Europe, Asia-Pacific, Latin America, Middle East and Africa), By Statistics, Trends, Outlook and Forecast 2026 to 2033 (Financial Impact Analysis)
ID : MRU_ 443815 | Date : Feb, 2026 | Pages : 248 | Region : Global | Publisher : MRU
Medical Disposable Gloves Market Size
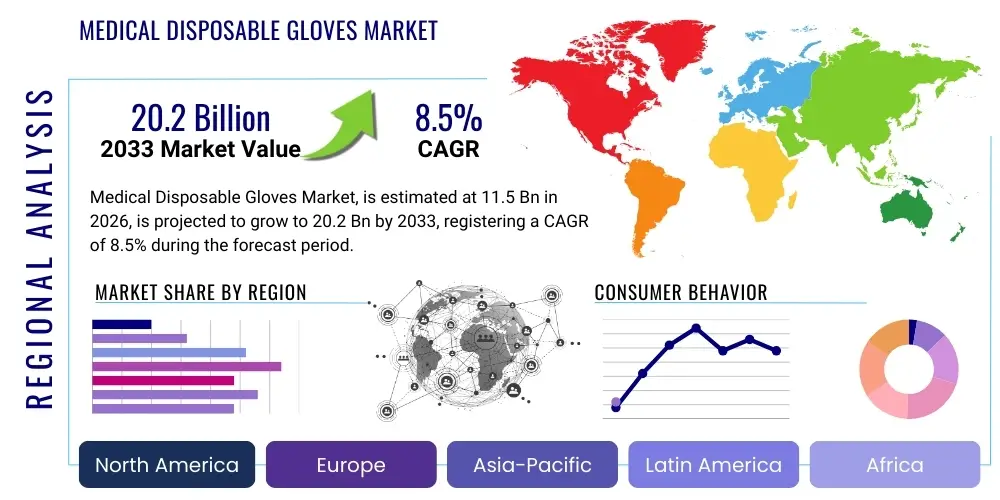
The Medical Disposable Gloves Market is projected to grow at a Compound Annual Growth Rate (CAGR) of 8.5% between 2026 and 2033. The market is estimated at USD 11.5 Billion in 2026 and is projected to reach USD 20.2 Billion by the end of the forecast period in 2033. This substantial expansion is fundamentally driven by the escalating global focus on infection prevention, rigorous enforcement of occupational safety standards in healthcare settings, and the continuous expansion of the global healthcare infrastructure, particularly in emerging economies where medical access is rapidly improving.
Medical Disposable Gloves Market introduction
The Medical Disposable Gloves Market encompasses specialized barrier protective equipment designed for single use in clinical and laboratory environments to prevent cross-contamination between caregivers and patients, or between potentially hazardous materials and the user. These gloves, critical for maintaining aseptic conditions, are primarily manufactured from materials such as nitrile rubber, natural rubber latex, vinyl (PVC), and increasingly, advanced synthetic polymers like neoprene. The fundamental utility of disposable medical gloves spans across diagnostic procedures, surgical interventions, general patient care, and handling of chemotherapy agents or infectious waste, cementing their role as an indispensable component of global biosafety protocols and standard healthcare practice.
The core product description includes both examination gloves, which are suitable for most non-surgical tasks requiring basic barrier protection, and surgical gloves, which demand higher quality standards concerning tactile sensitivity, fit, and sterility, often requiring rigorous testing and certification (such as FDA Class I or II). Major applications are broad, ranging from large hospital systems and specialized surgical centers to small clinics, dental offices, and long-term care facilities. The paramount benefits derived from their use include superior infection control, minimization of occupational exposure risks, and compliance with stringent public health regulations. The widespread adoption is further amplified by driving factors such as heightened awareness of hospital-acquired infections (HAIs), the rapid global spread of contagious diseases requiring continuous protection, and increasing healthcare expenditure that supports the routine use of high-quality disposable protective gear.
Furthermore, the market's trajectory is deeply influenced by shifts in material preference, specifically the continued movement away from natural rubber latex due to allergy concerns toward synthetic alternatives, predominantly nitrile. Nitrile gloves offer superior puncture resistance, chemical impermeability, and increasingly competitive pricing, positioning them as the material of choice for demanding medical environments. The innovation cycle within this industry is focused on developing gloves with enhanced comfort, reduced environmental impact through biodegradable formulations, and integrated anti-microbial properties, all aiming to optimize user compliance and overall clinical efficacy.
Medical Disposable Gloves Market Executive Summary
The Medical Disposable Gloves Market is experiencing robust growth characterized by intense competition and significant supply chain restructuring. Current business trends indicate a concentrated effort by major manufacturers, primarily based in Southeast Asia, to enhance production capacities through automation and vertical integration, aiming to mitigate the risks associated with volatile raw material prices, particularly for nitrile butadiene rubber (NBR). A noticeable trend involves strategic mergers and acquisitions focused on securing distribution networks in key consumption regions like North America and Europe, alongside heavy investment in research for sustainable and biodegradable glove options to address mounting environmental concerns and evolving consumer preferences. Furthermore, market players are increasingly segmenting their offerings to cater specifically to high-risk environments (e.g., chemotherapy gloves) versus general examination needs, maximizing margin capture across varied clinical applications.
Regionally, the market exhibits a clear bifurcation: Asia Pacific (APAC) remains the dominant manufacturing hub, leveraging lower operational costs and established polymer supply chains to fuel global supply. However, North America and Europe maintain their status as the largest revenue generators due to high per-capita healthcare spending, stringent regulatory environments mandating glove use, and high adoption rates of premium synthetic gloves. Specific regional trends include increasing domestic manufacturing initiatives in North America spurred by supply chain disruption resilience planning, and sustained regulatory pressure in Europe regarding chemical residues and quality control, leading to premiumization in those markets. The Middle East and Africa (MEA) and Latin America (LATAM) are emerging as high-growth potential regions, driven by significant government investments in healthcare infrastructure and increasing awareness of standardized infection control measures.
In terms of segment trends, the Nitrile segment is overwhelmingly dominant and projected to exhibit the highest CAGR, largely replacing latex in non-surgical and many surgical applications due to superior performance and hypoallergenic properties. The Examination Gloves category retains the largest market share by volume, necessitated by the sheer frequency of routine clinical procedures. Meanwhile, the Surgical Gloves segment, while smaller in volume, commands a higher average selling price due to the necessity for specialized barrier protection, sterility assurance, and enhanced tactile sensitivity required in operating theaters. End-user trends show hospitals and clinics maintaining their position as the primary consumption hubs, although growth in the ambulatory surgical centers (ASCs) and diagnostic laboratories segments is accelerating rapidly, reflecting the decentralized nature of modern healthcare delivery.
AI Impact Analysis on Medical Disposable Gloves Market
User queries regarding the impact of Artificial Intelligence (AI) on the Medical Disposable Gloves Market frequently revolve around three core themes: optimizing manufacturing efficiency, enhancing product quality and safety, and improving supply chain resilience and forecasting capabilities. Users are keen to understand if AI can significantly reduce production costs, specifically through predictive maintenance of high-speed manufacturing lines, thereby increasing yield and reducing waste. A major concern is how AI can be deployed in advanced quality control systems, ensuring zero-defect rates in critical products like surgical gloves, possibly through advanced computer vision systems for microscopic defect detection. Expectations also center on AI’s role in managing highly volatile raw material procurement and demand forecasting, especially in anticipation of future pandemic events or seasonal outbreaks, thereby stabilizing market supply and pricing, which has historically been erratic.
- AI-driven predictive maintenance minimizes production line downtime, optimizing overall equipment effectiveness (OEE) and reducing manufacturing costs.
- Advanced machine vision systems employing deep learning algorithms enable high-speed, non-destructive, and precise quality inspection, ensuring superior defect detection (pinholes, micro-tears) in gloves.
- AI-enhanced supply chain management provides real-time visibility and predictive demand forecasting, optimizing inventory levels of critical raw materials like NBR and stabilizing procurement strategies against global volatility.
- Integration of AI in material science research accelerates the discovery and testing of new, highly efficient, and biodegradable polymer formulations suitable for medical-grade disposable gloves.
- AI analytics support regulatory compliance tracking by monitoring batch quality data and generating automated reports, streamlining certification processes for global markets.
DRO & Impact Forces Of Medical Disposable Gloves Market
The market dynamics are governed by a complex interplay of Drivers, Restraints, and Opportunities (DRO), which collectively constitute the Impact Forces shaping the industry's evolution. Key drivers propelling the market include the mandatory enforcement of stringent infection control guidelines by global health organizations (WHO, CDC), the rapid expansion of the geriatric population requiring consistent medical care, and continuous investment in healthcare infrastructure across developing economies. These drivers ensure sustained, high-volume demand. However, the market faces significant restraints, notably the acute volatility and dependence on fossil-fuel-derived raw material prices (NBR), increasing scrutiny over environmental waste associated with single-use plastics, and global shortages of skilled labor in highly specialized manufacturing facilities, constraining capacity expansion. These constraints exert upward pressure on operational costs and limit market stability. Opportunities are abundant, focusing on innovation in biodegradable materials (e.g., polyurethanes, polylactic acid blends) as a sustainable alternative, penetrating highly underserved rural healthcare markets, and developing specialized gloves with enhanced features like antiviral or anti-fungal coatings. The collective impact forces result in a market characterized by high regulatory hurdles, consistent baseline growth due to indispensable product utility, and intense competitive focus on vertical integration and sustainable innovation to secure future growth pathways.
Segmentation Analysis
The Medical Disposable Gloves Market is comprehensively segmented based on material, application, and end-user, providing a granular view of market dynamics and consumer preferences across different clinical settings. Material segmentation is crucial as it dictates performance characteristics, cost, and hypoallergenic status, with Nitrile leading the charge due to its balance of strength and chemical resistance. Application segmentation distinguishes between non-invasive routine examinations and highly specialized surgical procedures, reflecting varying quality and regulatory requirements. End-user analysis identifies major consumption hubs, such as large tertiary care hospitals, which necessitate massive volumes, and high-growth segments like ambulatory care centers and home healthcare settings, where decentralized demand is escalating. Understanding these segments is vital for manufacturers to tailor their product offerings, pricing strategies, and distribution channels effectively.
- Material:
- Natural Rubber Latex
- Nitrile Rubber
- Vinyl/PVC
- Polyisoprene
- Neoprene and Others
- Application:
- Surgical
- Examination (Non-Surgical)
- End-User:
- Hospitals and Clinics
- Ambulatory Surgical Centers (ASCs)
- Diagnostic and Imaging Laboratories
- Pharmaceutical and Biotechnology Industries
- Research and Academic Institutions
- Dental Clinics
- Home Healthcare Settings
- Product Type:
- Powdered
- Non-Powdered
Value Chain Analysis For Medical Disposable Gloves Market
The value chain for medical disposable gloves is long and complex, beginning with upstream activities dominated by petrochemical and natural rubber suppliers. Upstream analysis focuses on the sourcing of critical raw materials, predominantly synthetic polymers like Nitrile Butadiene Rubber (NBR) latex and PVC resins, alongside natural rubber latex. Price stability and consistent quality of these raw materials are primary determinants of manufacturing profitability. The subsequent manufacturing phase involves capital-intensive, high-speed dipping and curing processes, predominantly centralized in Southeast Asia (Malaysia, Thailand, China). Efficiency gains in this stage, driven by automation and energy recovery systems, are crucial for competitive pricing. Midstream activities involve stringent quality control checks, sterilization processes for surgical gloves, packaging, and regulatory certification (FDA, CE marking), which add significant value and cost.
The downstream segment encompasses the distribution network, logistics, and end-user interaction. Distribution channels are highly varied, often utilizing large, specialized healthcare distributors (e.g., McKesson, Owens & Minor) who manage inventory and logistics for hospitals and large clinic networks. Direct sales channels are employed primarily for very large institutional contracts or highly specialized, high-margin products. The reliance on indirect distribution allows manufacturers to leverage the extensive warehousing and delivery infrastructure of third-party logistics providers. Final delivery to end-users (hospitals, clinics) requires sophisticated just-in-time inventory management to ensure continuous supply, especially given the non-negotiable nature of demand for infection control products. The efficiency of the distribution system directly impacts the availability and final cost of the gloves to the healthcare provider.
The complexity of the global supply chain, recently highlighted by pandemic-related disruptions, necessitates robust inventory strategies and geographical diversification. Manufacturers are increasingly integrating forward through acquiring or establishing dedicated distribution centers in major consumption markets to reduce lead times and buffer against geopolitical risks. Furthermore, transparency and traceability throughout the value chain are becoming paramount due to increasing regulatory and ethical scrutiny regarding sourcing practices, particularly concerning sustainable rubber harvesting and labor standards in manufacturing facilities.
Medical Disposable Gloves Market Potential Customers
The primary consumers and end-users of medical disposable gloves are institutions and professionals within the comprehensive healthcare ecosystem where barrier protection is mandatory for clinical interaction and material handling. Hospitals, including general, specialty, and teaching hospitals, represent the largest volume purchasers, driven by the requirement for massive inventories across emergency rooms, operating theaters, intensive care units, and general patient wards. Closely following are clinics and physicians’ offices, which utilize high volumes of examination gloves for routine patient interactions and minor procedures. The increasing shift towards outpatient care has amplified the importance of Ambulatory Surgical Centers (ASCs), which require sterile surgical gloves and specialized examination gloves for various day procedures.
Beyond traditional patient care settings, diagnostic and imaging laboratories constitute significant end-users, where gloves are essential for handling clinical specimens, testing reagents, and ensuring environmental safety. The Pharmaceutical and Biotechnology sectors utilize specialized disposable gloves extensively, particularly in manufacturing cleanrooms, R&D labs, and quality control departments where contamination prevention is mission-critical. Furthermore, the burgeoning segment of home healthcare providers and long-term care facilities represents a rapidly growing customer base, driven by the aging population and the decentralization of chronic care management, where gloves are necessary for maintaining hygienic practices outside institutional settings.
Lastly, governmental organizations, including national stockpiles and public health agencies, act as strategic buyers, particularly during periods of high infectious disease risk, maintaining reserves to ensure national health security. The procurement decisions of these diverse customer groups are influenced by a combination of factors: price, material type (allergy management), regulatory compliance, and increasingly, sustainability credentials and supplier reliability, demanding that manufacturers offer a differentiated product portfolio capable of meeting specialized needs across the entire spectrum of clinical applications.
| Report Attributes | Report Details |
|---|---|
| Market Size in 2026 | USD 11.5 Billion |
| Market Forecast in 2033 | USD 20.2 Billion |
| Growth Rate | 8.5% CAGR |
| Historical Year | 2019 to 2024 |
| Base Year | 2025 |
| Forecast Year | 2026 - 2033 |
| DRO & Impact Forces |
|
| Segments Covered |
|
| Key Companies Covered | Ansell Limited, Top Glove Corporation Bhd, Hartalega Holdings Berhad, Supermax Corporation Berhad, Kossan Rubber Industries Bhd, Medline Industries, Inc., Cardinal Health, Inc., Dynarex Corporation, Kimberly-Clark Corporation, Semperit AG Holding, 3M Company, Adventa Berhad, Molnlycke Health Care AB, Shield Scientific, Unigloves GmbH, Rubberex Corporation, Tronex International, Inc., Safeskin Corporation, WRP Asia Pacific, Inc., Careplus Group Berhad. |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Enquiry Before Buy | Have specific requirements? Send us your enquiry before purchase to get customized research options. Request For Enquiry Before Buy |
Medical Disposable Gloves Market Key Technology Landscape
The Medical Disposable Gloves Market is characterized by continuous technological refinement aimed at improving barrier integrity, user comfort, sustainability, and production efficiency. A critical technology involves advanced polymerization techniques used in the production of synthetic rubber, specifically modifying Nitrile Butadiene Rubber (NBR) latex formulations to enhance elasticity and tactile sensitivity, bridging the gap between nitrile and natural rubber performance. Manufacturers are employing proprietary compounding techniques to reduce the thickness of nitrile gloves without compromising tensile strength or puncture resistance, thereby reducing material usage and costs while enhancing dexterity for complex surgical procedures. Furthermore, innovations in chlorination and polymer coating technologies are key to creating non-stick, donning-friendly gloves that eliminate the need for allergy-inducing powder.
A significant area of focus is the integration of anti-microbial and antiviral coating technologies. These surface treatments, applied during the curing or post-processing stages, aim to actively reduce pathogens on the glove surface, providing an added layer of infection control beyond the basic barrier function. While still maturing, these functionalized gloves represent the next generation of high-performance PPE, particularly valuable in high-risk infection environments. Automation technology is also transforming the manufacturing landscape; high-throughput automated dipping lines, coupled with robotic handling and sophisticated quality monitoring, are crucial for maintaining high volumes and consistent quality, mitigating labor costs, and ensuring energy efficiency through improved process control.
Finally, technology related to environmental sustainability is gaining traction. Research and development efforts are heavily vested in creating truly biodegradable or compostable glove materials, often leveraging polymers derived from plant-based sources or specialized synthetic compounds designed to degrade quickly in industrial composting settings. Simultaneously, manufacturing facilities are adopting advanced water treatment and air filtration systems to minimize the environmental footprint associated with latex dipping and chemical processing, responding proactively to global calls for responsible manufacturing practices within the medical supply industry.
Regional Highlights
- North America (NA): Represents a mature and high-value market, characterized by extremely strict regulatory standards (FDA compliance) and high per-capita healthcare expenditure. The demand is heavily skewed towards premium non-latex alternatives, particularly nitrile and specialized surgical gloves. Recent focus has been on securing resilient domestic supply chains following pandemic-induced shortages, leading to increased governmental contracts and investment in localized manufacturing capabilities, albeit at a higher production cost compared to Asia.
- Europe: Driven by an aging population, universal healthcare coverage, and rigorous occupational health and safety regulations (e.g., MDR compliance). Germany, the UK, and France are the largest consuming nations. The European market places a strong emphasis on sustainability and corporate social responsibility (CSR) in procurement, favoring suppliers who demonstrate ethical sourcing and provide low-protein or sustainable material options.
- Asia Pacific (APAC): Dominates the global market both in terms of manufacturing capacity and overall volume consumption growth. Countries like Malaysia, Thailand, and China are global production hubs for natural and synthetic latex gloves. Consumption growth is skyrocketing due to rapid expansion of healthcare infrastructure, increasing population density, and rising awareness regarding disease prevention in populous countries like India and China, transitioning rapidly from reusable to disposable practices.
- Latin America (LATAM): Exhibits significant growth potential fueled by improving economic conditions, urbanization, and increasing access to formal healthcare services. Brazil and Mexico are key markets, though the region often faces challenges related to pricing sensitivity and reliance on imports, which can be susceptible to currency fluctuations and trade tariffs, driving demand primarily for cost-effective vinyl and standard examination gloves.
- Middle East and Africa (MEA): A rapidly emerging market segment, particularly in the Gulf Cooperation Council (GCC) states (UAE, Saudi Arabia) due to high government spending on world-class healthcare facilities and medical tourism initiatives. Africa's demand is primarily driven by international aid programs and efforts to combat endemic infectious diseases, resulting in high variability in quality requirements and price points across the continent.
Top Key Players
The market research report includes a detailed profile of leading stakeholders in the Medical Disposable Gloves Market.- Ansell Limited
- Top Glove Corporation Bhd
- Hartalega Holdings Berhad
- Supermax Corporation Berhad
- Kossan Rubber Industries Bhd
- Medline Industries, Inc.
- Cardinal Health, Inc.
- Dynarex Corporation
- Kimberly-Clark Corporation
- Semperit AG Holding
- 3M Company
- Adventa Berhad
- Molnlycke Health Care AB
- Shield Scientific
- Unigloves GmbH
- Rubberex Corporation
- Tronex International, Inc.
- Safeskin Corporation
- WRP Asia Pacific, Inc.
- Careplus Group Berhad
Frequently Asked Questions
Analyze common user questions about the Medical Disposable Gloves market and generate a concise list of summarized FAQs reflecting key topics and concerns.What is driving the transition from Latex to Nitrile gloves in the medical sector?
The transition is primarily driven by increasing awareness and prevalence of Type I allergic reactions associated with natural rubber latex proteins. Nitrile gloves offer superior chemical resistance, puncture strength, and are inherently protein-free and powder-free, making them the preferred, hypoallergenic, and high-performance standard for critical medical procedures.
How significant are sustainability concerns in the disposable gloves market?
Sustainability is a rapidly increasing concern, as disposable gloves contribute vast quantities of medical waste to landfills globally. This drives market opportunities for manufacturers investing heavily in developing and commercializing biodegradable, compostable, or alternative material gloves that minimize long-term environmental impact without compromising barrier efficacy.
Which geographical region dominates the manufacturing of medical disposable gloves?
The Asia Pacific (APAC) region, specifically countries like Malaysia, Thailand, and China, overwhelmingly dominates global manufacturing capacity due to abundant raw material supplies (both natural and synthetic latex bases), established manufacturing infrastructure, and lower operational costs, making it the primary hub for global supply.
What is the primary difference between surgical gloves and examination gloves?
Surgical gloves are sterilized, meet higher regulatory standards (e.g., concerning Acceptable Quality Level or AQL), are anatomically shaped, and offer enhanced tactile sensitivity and longer cuffs for use in sterile operating environments. Examination gloves are non-sterile (though some are available sterile) and designed for general patient care and routine non-invasive procedures.
How does the volatility of raw material prices affect the market?
Raw material price volatility, particularly for Nitrile Butadiene Rubber (NBR), significantly impacts manufacturer profitability and final product pricing. Because the manufacturing process is highly dependent on petrochemical derivatives, global oil price fluctuations and supply chain disruptions directly influence the cost of synthetic gloves, leading to periodic price instability for healthcare providers.
To check our Table of Contents, please mail us at: sales@marketresearchupdate.com
Research Methodology
The Market Research Update offers technology-driven solutions and its full integration in the research process to be skilled at every step. We use diverse assets to produce the best results for our clients. The success of a research project is completely reliant on the research process adopted by the company. Market Research Update assists its clients to recognize opportunities by examining the global market and offering economic insights. We are proud of our extensive coverage that encompasses the understanding of numerous major industry domains.
Market Research Update provide consistency in our research report, also we provide on the part of the analysis of forecast across a gamut of coverage geographies and coverage. The research teams carry out primary and secondary research to implement and design the data collection procedure. The research team then analyzes data about the latest trends and major issues in reference to each industry and country. This helps to determine the anticipated market-related procedures in the future. The company offers technology-driven solutions and its full incorporation in the research method to be skilled at each step.
The Company's Research Process Has the Following Advantages:
- Information Procurement
The step comprises the procurement of market-related information or data via different methodologies & sources.
- Information Investigation
This step comprises the mapping and investigation of all the information procured from the earlier step. It also includes the analysis of data differences observed across numerous data sources.
- Highly Authentic Source
We offer highly authentic information from numerous sources. To fulfills the client’s requirement.
- Market Formulation
This step entails the placement of data points at suitable market spaces in an effort to assume possible conclusions. Analyst viewpoint and subject matter specialist based examining the form of market sizing also plays an essential role in this step.
- Validation & Publishing of Information
Validation is a significant step in the procedure. Validation via an intricately designed procedure assists us to conclude data-points to be used for final calculations.
×
Request Free Sample:
Related Reports
Select License

Why Choose Us

We're cost-effective and Offered Best services:
We are flexible and responsive startup research firm. We adapt as your research requires change, with cost-effectiveness and highly researched report that larger companies can't match.

Information Safety
Market Research Update ensure that we deliver best reports. We care about the confidential and personal information quality, safety, of reports. We use Authorize secure payment process.

We Are Committed to Quality and Deadlines
We offer quality of reports within deadlines. We've worked hard to find the best ways to offer our customers results-oriented and process driven consulting services.

Our Remarkable Track Record
We concentrate on developing lasting and strong client relationship. At present, we hold numerous preferred relationships with industry leading firms that have relied on us constantly for their research requirements.

Best Service Assured
Buy reports from our executives that best suits your need and helps you stay ahead of the competition.

Customized Research Reports
Our research services are custom-made especially to you and your firm in order to discover practical growth recommendations and strategies. We don't stick to a one size fits all strategy. We appreciate that your business has particular research necessities.

Service Assurance
At Market Research Update, we are dedicated to offer the best probable recommendations and service to all our clients. You will be able to speak to experienced analyst who will be aware of your research requirements precisely.
Contact With Our Sales Team
Customer Testimonials
The content of the report is always up to the mark. Good to see speakers from expertise authorities.
Privacy requested , Managing Director
A lot of unique and interesting topics which are described in good manner.
Privacy requested, President
Well researched, expertise analysts, well organized, concrete and current topics delivered in time.
Privacy requested, Development Manager