Ferric Citrate Market Size, By Region (North America, Europe, Asia-Pacific, Latin America, Middle East and Africa), By Statistics, Trends, Outlook and Forecast 2026 to 2033 (Financial Impact Analysis)
ID : MRU_ 443666 | Date : Feb, 2026 | Pages : 255 | Region : Global | Publisher : MRU
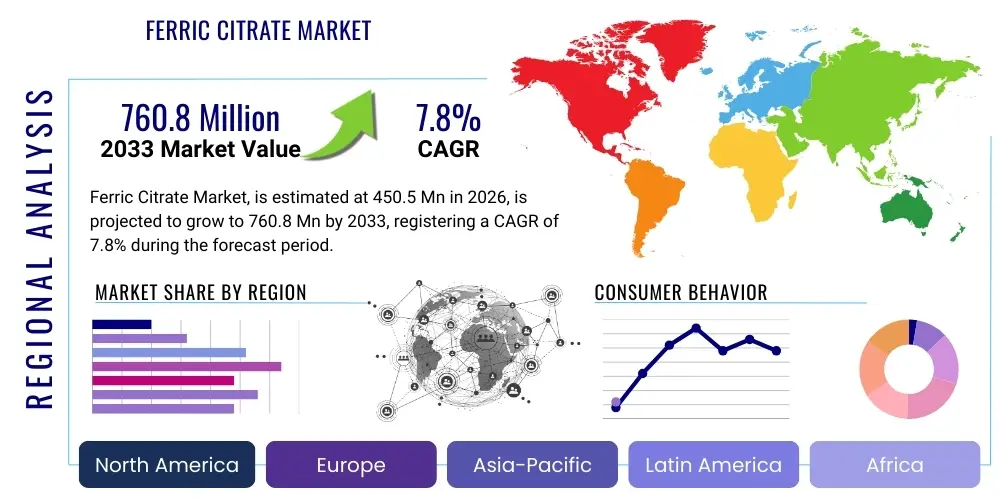
Ferric Citrate Market Size
The Ferric Citrate Market is projected to grow at a Compound Annual Growth Rate (CAGR) of 7.8% between 2026 and 2033. The market is estimated at USD 450.5 Million in 2026 and is projected to reach USD 760.8 Million by the end of the forecast period in 2033. This consistent expansion is predominantly driven by the increasing global prevalence of Chronic Kidney Disease (CKD) and the subsequent rise in patients requiring treatment for associated complications such as hyperphosphatemia and iron deficiency anemia (IDA). The introduction of improved formulations and broader geographical approvals, particularly in emerging Asian markets, are critical factors fueling this robust market trajectory. Furthermore, heightened patient awareness regarding managing mineral and bone disorders (MBD) related to CKD contributes significantly to the anticipated market value appreciation.
Ferric Citrate Market introduction
The Ferric Citrate Market encompasses pharmaceutical-grade products primarily utilized for the management of hyperphosphatemia, a common complication among Chronic Kidney Disease (CKD) patients undergoing dialysis, and for treating iron deficiency anemia (IDA) in non-dialysis-dependent CKD patients. Ferric citrate acts as an iron-based phosphate binder in the gastrointestinal tract, forming insoluble complexes with dietary phosphate, thereby preventing its absorption and controlling serum phosphorus levels. Simultaneously, the iron component is absorbed systemically, contributing to increased iron stores and hemoglobin levels, offering a dual therapeutic benefit highly valued in nephrology. This unique mechanism positions ferric citrate as a crucial therapeutic agent, offering efficacy and convenience compared to older generation binders.
The principal applications of ferric citrate span clinical nephrology and hematology. In hyperphosphatemia management, it addresses the critical need to control serum phosphate to prevent secondary hyperparathyroidism, renal osteodystrophy, and cardiovascular complications, which are major causes of morbidity and mortality in the CKD population. For IDA treatment in non-dialysis CKD patients, its oral administration offers a viable alternative to intravenous iron, reducing logistical burdens and potential side effects associated with infusion centers. The product's stability, high efficacy profile, and demonstrated clinical outcomes in reducing the need for intravenous iron and erythropoiesis-stimulating agents (ESAs) have firmly established its market presence globally. Regulatory approvals across major economies, including the U.S. (as Auryxia) and specific European and Asian countries, underscore its therapeutic importance.
Key benefits driving the adoption of ferric citrate include its dual mechanism of action, which simplifies medication regimens for complex CKD patients, and its superior efficacy in lowering phosphate levels while concurrently improving iron parameters. The market is also propelled by the global increase in the incidence of lifestyle diseases such as diabetes and hypertension, which are primary precursors to CKD. Furthermore, continuous investments in research and development aimed at improving patient compliance through novel dosing schedules and enhanced palatability are expected to sustain market growth. However, market penetration is influenced by stringent regulatory processes and price competition from generic alternatives and existing phosphate binders.
Ferric Citrate Market Executive Summary
The Ferric Citrate Market is characterized by strong demand originating from the expanding global burden of Chronic Kidney Disease (CKD), particularly in regions witnessing rapid aging populations and increasing prevalence of comorbidities like diabetes. Key business trends include strategic collaborations between pharmaceutical companies and specialized nephrology centers to enhance product visibility and prescribing rates. There is a noticeable shift toward optimizing patient compliance through fixed-dose combinations and digital adherence programs. Manufacturers are also focusing on evidence-based marketing, leveraging results from large-scale Phase IV clinical trials demonstrating long-term safety and efficacy, which is crucial for securing preferred formulary status with payers and healthcare providers, ensuring sustained revenue streams in competitive environments.
Regionally, North America remains the leading market owing to established reimbursement policies, high CKD prevalence, and rapid adoption of novel pharmaceutical treatments. However, the Asia Pacific (APAC) region is projected to exhibit the fastest growth rate, fueled by improving healthcare infrastructure, increasing diagnostic rates for CKD, and governmental initiatives aimed at expanding access to essential medicines. Europe maintains a steady growth trajectory, influenced by centralized drug procurement policies and varying national reimbursement schemes. The competitive landscape is intensely focused on patent exclusivity maintenance and the timely filing of supplemental New Drug Applications (sNDAs) to expand approved indications, thereby maximizing the return on substantial research investments.
Segmentation trends highlight the dominance of the hyperphosphatemia treatment segment due to the large patient pool requiring continuous phosphate management. Within the distribution channels, hospital pharmacies and specialty clinics account for the largest share, reflecting the necessity of specialized nephrology consultation for treatment initiation. The oral tablet formulation remains the preferred product type, emphasizing ease of use and standardization. Looking ahead, the market anticipates sustained innovation in drug delivery systems and formulation stability to further enhance patient outcomes and broaden therapeutic applicability across the entire spectrum of CKD progression, from early-stage management to end-stage renal disease (ESRD).
AI Impact Analysis on Ferric Citrate Market
User inquiries regarding the impact of Artificial Intelligence (AI) on the Ferric Citrate market primarily center on optimizing drug synthesis, accelerating clinical trial enrollment and analysis for new indications, and refining patient stratification for personalized dosing. Users are keen to understand how AI-driven predictive modeling can improve adherence rates among CKD patients, a significant challenge in chronic disease management. Key concerns revolve around the integration of AI tools into existing pharmaceutical manufacturing processes without compromising stringent regulatory standards and ensuring data security in handling sensitive patient records collected through AI-enabled monitoring systems. Expectations include AI leading to cost reduction in clinical development and facilitating the discovery of synergistic therapeutic uses for ferric citrate beyond its current indications, potentially expanding its market application and improving overall resource efficiency across the product lifecycle.
The application of AI in the manufacturing process of ferric citrate involves optimizing reaction kinetics and crystallization techniques to ensure high purity and yield, critical factors in pharmaceutical production. Machine learning algorithms can analyze vast datasets from bioreactors and chemical synthesis units to predict optimal operational parameters, minimizing waste and ensuring batch-to-batch consistency, thereby addressing strict Current Good Manufacturing Practice (cGMP) requirements. Furthermore, AI-powered quality control systems using image recognition and spectroscopic analysis can rapidly detect contaminants or inconsistencies, significantly reducing the turnaround time for quality assurance and improving the overall supply chain reliability of the drug, which is essential for chronic therapies.
In the clinical sphere, AI is transformative in identifying suitable patient populations for clinical trials, particularly for exploring ferric citrate's role in new therapeutic areas, such as specific subsets of non-CKD anemia or inflammatory conditions where iron metabolism is impaired. Natural Language Processing (NLP) tools can rapidly screen electronic health records (EHRs) to identify patients matching complex inclusion/exclusion criteria, drastically reducing enrollment time. Moreover, AI predictive models can analyze patient response data (serum phosphate, ferritin, hemoglobin) in real-time, allowing clinicians to adjust dosage proactively, leading to more personalized treatment regimens and potentially better clinical outcomes, thus strengthening ferric citrate’s competitive profile against other phosphate binders and iron supplements.
- Enhanced predictive modeling for CKD progression and IDA risk assessment.
- Optimization of chemical synthesis routes to reduce manufacturing costs and variability.
- Acceleration of clinical trial patient recruitment and outcome analysis using NLP on EHR data.
- Development of AI-driven patient adherence monitoring programs and personalized dosing algorithms.
- Automated quality control and impurity detection in bulk drug substance production.
- Identification of novel, secondary therapeutic targets for ferric citrate through deep learning analysis of biological pathways.
DRO & Impact Forces Of Ferric Citrate Market
The Ferric Citrate Market dynamics are shaped by a complex interplay of Drivers, Restraints, and Opportunities (DRO). The primary driving force is the escalating global prevalence of CKD, directly increasing the patient pool requiring phosphate binders and iron supplementation. Furthermore, clinical guidelines increasingly advocate for effective management of both hyperphosphatemia and IDA in CKD patients, providing a strong clinical imperative for dual-action agents like ferric citrate. Opportunities emerge from expanding market penetration into underserved geographies, notably parts of Latin America and Southeast Asia, where CKD incidence is rising but specialized treatment access is limited. Additionally, exploring non-dialysis dependent CKD indications and broadening its use in managing chronic iron deficiency in other complex patient groups represent significant future growth avenues. Technological advancements in formulation science to improve patient compliance also serve as key opportunities, mitigating some of the traditional challenges associated with chronic oral medication regimens.
Restraints impeding aggressive market growth include the high cost associated with proprietary ferric citrate formulations compared to generic aluminum or calcium-based phosphate binders, placing pressure on healthcare budgets, particularly in price-sensitive markets. Regulatory hurdles, especially in obtaining approvals for pediatric use or new geographic regions, require extensive investment and time. Furthermore, the market faces significant competition from established non-iron phosphate binders (e.g., sevelamer and lanthanum carbonate), which have strong prescriber loyalty and robust clinical track records. Patient non-adherence due to pill burden and potential gastrointestinal side effects remains a perennial challenge affecting overall treatment effectiveness and requiring continuous patient education efforts.
The Impact Forces shaping the market include technological advancements that reduce production costs, making ferric citrate more economically viable globally. The shift in global demographics toward older populations with higher rates of CKD acts as a long-term, powerful driver. Conversely, changes in reimbursement policies or the listing of generic competitors in major markets (e.g., U.S. and Europe) constitute significant restraining forces capable of rapidly altering market share. The clinical efficacy demonstrated through robust real-world evidence (RWE) studies, affirming the drug's dual benefit, enhances its clinical acceptance and acts as a positive impact force, strengthening prescriber confidence and overcoming initial reluctance related to novel therapeutic class introduction.
Segmentation Analysis
The Ferric Citrate market is strategically segmented based on application, end-user, and distribution channel, reflecting the diverse clinical settings where the product is utilized. Analyzing these segments provides nuanced insights into demand patterns and competitive intensity across different market niches. The segmentation based on application, specifically hyperphosphatemia versus iron deficiency anemia (IDA) treatment, is critical, as the reimbursement landscape and patient populations for these two indications vary significantly, although the product often serves both needs simultaneously in a CKD context. Understanding the dominant end-user segments, primarily hospitals and specialized nephrology centers, is essential for designing targeted marketing and distribution strategies that address the specific needs of healthcare professionals managing complex renal patients.
From a product type perspective, the segmentation primarily focuses on the solid oral dosage form (tablets or capsules), which currently dominates the market due to convenience and established manufacturing practices. Future segmentation might incorporate extended-release formulations or liquid dosage forms aimed at pediatric populations or patients with swallowing difficulties, responding to unmet clinical needs. Geographic segmentation remains paramount, with North America and Europe defining current revenue strength, while the burgeoning patient population and improving healthcare access in the Asia Pacific region drive future growth projections, necessitating distinct regulatory and commercialization approaches tailored to regional nuances.
The depth of market analysis relies on segment performance evaluations to identify high-growth areas. The IDA segment in non-dialysis CKD patients, while smaller than hyperphosphatemia in dialysis patients, offers higher growth potential due to its preventive therapeutic role earlier in the disease progression and less competition from traditional non-iron binders. The distribution channel segment reveals the continued importance of direct sales forces targeting nephrology specialists, underscoring the specialty nature of the drug and the need for clinical education to ensure appropriate prescribing practices, particularly regarding monitoring iron parameters alongside phosphate levels.
- By Application:
- Hyperphosphatemia in Dialysis-Dependent CKD Patients
- Iron Deficiency Anemia (IDA) in Non-Dialysis Dependent CKD Patients
- By End-User:
- Hospitals
- Nephrology Clinics and Dialysis Centers
- Retail and Online Pharmacies
- By Formulation:
- Tablets (Oral Solid Dosage)
- Capsules (Oral Solid Dosage)
- By Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- By Region:
- North America (U.S., Canada)
- Europe (Germany, U.K., France, Italy, Spain, Rest of Europe)
- Asia Pacific (China, Japan, India, South Korea, Rest of APAC)
- Latin America (Brazil, Mexico, Rest of LATAM)
- Middle East and Africa (GCC Countries, South Africa, Rest of MEA)
Value Chain Analysis For Ferric Citrate Market
The value chain for the Ferric Citrate Market begins with the rigorous upstream process involving the procurement of raw materials, chiefly pharmaceutical-grade iron salts and citric acid derivatives. This stage requires meticulous supplier qualification and stringent quality control, as the purity of the intermediates directly impacts the final product efficacy and safety profile. Chemical synthesis and purification, typically outsourced to highly specialized Active Pharmaceutical Ingredient (API) manufacturers, constitute the core of the upstream segment. Due to the complex nature of chelating iron and citrate for pharmaceutical use, robust intellectual property protection and advanced chemical engineering capabilities are crucial at this stage, setting a high barrier to entry for new competitors and ensuring product stability, which is vital for long-term storage and efficacy.
The midstream component encompasses formulation, dosage manufacturing, packaging, and regulatory approval. This stage is dominated by the branded pharmaceutical company holding the New Drug Application (NDA) or Marketing Authorization. Manufacturing facilities must adhere to global cGMP standards, especially those enforced by the FDA and EMA. Quality assurance and control checks are continuous to ensure uniformity in tablet strength and dissolution properties. The regulatory segment, while not physical, is intrinsically linked, involving continuous post-market surveillance and reporting, which adds significant overhead but maintains market access. Efficient manufacturing logistics, including facility utilization and minimizing batch variation, are key determinants of profitability in this mature stage of the product lifecycle.
The downstream activities involve distribution, marketing, and sales, leading directly to the end-user. Distribution channels are typically specialized, utilizing both direct sales forces targeting nephrology specialists and third-party specialty distributors who manage cold chain or controlled substance logistics. Direct distribution is favored in high-value markets (North America, Western Europe) to maintain margin control and gather direct market intelligence. Indirect channels, involving wholesalers and large pharmacy chains, ensure broad access. Marketing efforts are highly focused on presenting clinical evidence to nephrologists, dietitians, and dialysis center administrators. The high value of the treatment necessitates strong payer relations and sophisticated reimbursement management programs to ensure patient access and minimize out-of-pocket costs, completing the value delivery mechanism to the patient.
Ferric Citrate Market Potential Customers
The primary and most significant potential customers for ferric citrate are individuals suffering from Chronic Kidney Disease (CKD), specifically those requiring intervention for hyperphosphatemia or iron deficiency anemia (IDA). This demographic includes patients undergoing hemodialysis or peritoneal dialysis for End-Stage Renal Disease (ESRD), as hyperphosphatemia is nearly universal in this group due to compromised renal clearance. The second major customer segment comprises non-dialysis dependent CKD patients, particularly those in stages 3 to 5, who require proactive management of IDA to prevent complications and improve quality of life. These patient populations are identified and managed by specialized healthcare providers, making nephrology centers and affiliated dialysis units the key centers of consumption.
The secondary customer group includes the healthcare institutions and entities responsible for procuring and dispensing the medication. This encompasses hospital pharmacies, dedicated nephrology clinics, outpatient dialysis centers (both corporate and independent), and increasingly, specialized mail-order and online pharmacies catering to chronic disease patients. Procurement decisions at this level are heavily influenced by formulary inclusion, negotiated pricing (often involving large Group Purchasing Organizations or GPOs), inventory management capabilities, and successful navigation of complex insurance and reimbursement protocols. Therefore, manufacturers must align their sales and marketing efforts to address both the clinical prescriber and the institutional purchasing gatekeeper.
Emerging potential customer segments include patients with non-CKD related chronic iron malabsorption issues or individuals with specific genetic disorders affecting iron metabolism, provided that expanded indications are secured through further clinical trials and regulatory approvals. Furthermore, healthcare systems in developing economies, previously relying on older, less efficacious treatments, represent untapped customer potential as economic development and investment in specialized nephrology care increase. Targeting these diverse customer groups requires a multi-pronged strategy encompassing clinical data dissemination, pricing flexibility, and robust supply chain logistics ensuring product availability where specialized care is delivered.
| Report Attributes | Report Details |
|---|---|
| Market Size in 2026 | USD 450.5 Million |
| Market Forecast in 2033 | USD 760.8 Million |
| Growth Rate | 7.8% CAGR |
| Historical Year | 2019 to 2024 |
| Base Year | 2025 |
| Forecast Year | 2026 - 2033 |
| DRO & Impact Forces |
|
| Segments Covered |
|
| Key Companies Covered | Keryx Biopharmaceuticals (Akebia Therapeutics), Panion & BF Biotech Inc., Spectrum Chemical Manufacturing Corp., BASF SE, American Elements, Sisco Research Laboratories Pvt. Ltd., Merck KGaA, Avantor, Thermo Fisher Scientific, Finar Chemicals, Ferro Chem Industries, Jost Chemical, The Chemical Co., ProChem, Inc., Alfa Aesar (part of Thermo Fisher Scientific), TCI Chemicals, Glentham Life Sciences, Chem-Impex International. |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Enquiry Before Buy | Have specific requirements? Send us your enquiry before purchase to get customized research options. Request For Enquiry Before Buy |
Ferric Citrate Market Key Technology Landscape
The technology landscape for the Ferric Citrate market is primarily centered on advanced pharmaceutical formulation techniques and robust analytical methodologies ensuring product stability and bioavailability. A crucial technological element involves optimizing the synthesis process to achieve highly pure, amorphous or nano-crystalline forms of ferric citrate. This is vital because the active pharmaceutical ingredient (API) structure directly influences its binding capacity to phosphate in the gut and its subsequent iron release characteristics. Specialized spray drying or co-precipitation techniques are often employed to control particle size distribution, which affects dissolution rates and ensures optimal therapeutic efficacy while minimizing potential gastrointestinal irritation associated with unrefined iron salts.
Another significant technological advancement focuses on developing stable, high-density dosage forms that reduce the patient's daily pill burden. Since CKD patients often manage multiple chronic conditions, reducing the number of tablets required is paramount for improving compliance and overall treatment adherence, a key challenge in chronic renal care. Manufacturers are investing in advanced tableting machinery and excipient technologies that allow for higher API loading within smaller tablets while maintaining fast disintegration and controlled release profiles in the gastric environment. Furthermore, enteric coating technologies are being explored to modulate the release kinetics and absorption profile, optimizing the dual function of phosphate binding and systemic iron availability.
Beyond formulation, the technological landscape includes sophisticated analytical chemistry techniques, such as High-Performance Liquid Chromatography (HPLC) coupled with Mass Spectrometry (MS), for rigorous quality control of both the API and the finished product. These technologies are essential for detecting trace impurities and degradation products, ensuring the drug meets the stringent quality standards mandated by global regulatory bodies. Additionally, ongoing R&D focuses on continuous manufacturing processes, leveraging automation and process analytical technology (PAT) to enhance efficiency, reduce batch variability, and ultimately lower the cost of goods sold, positioning ferric citrate more competitively against generic alternatives in the market.
Regional Highlights
- North America (U.S. and Canada): North America is the leading revenue contributor, primarily due to the established infrastructure for CKD management, high per capita healthcare spending, and favorable reimbursement policies for branded specialty pharmaceuticals like ferric citrate. The U.S. market, driven by a high prevalence of ESRD and aggressive adoption of novel therapeutics through major dialysis providers, dictates global pricing and clinical adoption trends. Stringent FDA regulations ensure high product quality, while strong competition necessitates continuous investment in clinical efficacy studies and payer negotiation strategies.
- Europe (Germany, U.K., France, Italy, Spain): Europe represents a mature market characterized by universal healthcare systems and varied national reimbursement approaches. Adoption is strong, particularly in countries with high dialysis prevalence. However, pricing pressure from centralized health technology assessments (HTAs) often restrains market growth compared to the U.S. Focus is placed on comparative effectiveness research against existing phosphate binders (Sevelamer, Lanthanum) to justify premium pricing and maintain market access within the region's diverse regulatory landscape.
- Asia Pacific (APAC) (China, Japan, India): APAC is projected as the fastest-growing region, driven by the massive and rapidly increasing patient pool suffering from CKD, especially in populous nations like China and India, where diabetes and hypertension prevalence is soaring. Improving healthcare access, expanding insurance coverage, and local manufacturing initiatives are accelerating the adoption of advanced treatments. Japan, with its high geriatric population, remains a critical high-value market, while emerging economies focus on cost-effective alternatives and localized clinical data generation to support broader market uptake.
- Latin America (LATAM): LATAM markets, including Brazil and Mexico, exhibit significant potential, though growth is hampered by economic volatility and complex governmental procurement systems. Increasing awareness among nephrologists and efforts by multinational corporations to establish distribution networks are gradually improving market penetration. The emphasis is often on generating real-world evidence to support value propositions to overcome pricing challenges.
- Middle East and Africa (MEA): MEA presents a nascent market, with growth concentrated in the GCC countries (UAE, Saudi Arabia) due to advanced healthcare systems and high prevalence of lifestyle diseases leading to CKD. South Africa also shows steady adoption. Market challenges include the fragmented nature of healthcare delivery and reliance on imports, making robust supply chain management a key differentiator for companies operating in this region.
Top Key Players
The market research report includes a detailed profile of leading stakeholders in the Ferric Citrate Market.- Keryx Biopharmaceuticals (Akebia Therapeutics)
- Panion & BF Biotech Inc.
- Spectrum Chemical Manufacturing Corp.
- BASF SE
- American Elements
- Sisco Research Laboratories Pvt. Ltd.
- Merck KGaA
- Avantor
- Thermo Fisher Scientific
- Finar Chemicals
- Ferro Chem Industries
- Jost Chemical
- The Chemical Co.
- ProChem, Inc.
- Alfa Aesar (part of Thermo Fisher Scientific)
- TCI Chemicals
- Glentham Life Sciences
- Chem-Impex International
- Pfizer Inc. (Indirect presence through APIs)
- Teva Pharmaceutical Industries Ltd. (Potential generic entrants)
Frequently Asked Questions
Analyze common user questions about the Ferric Citrate market and generate a concise list of summarized FAQs reflecting key topics and concerns.What is the primary therapeutic application of Ferric Citrate?
Ferric citrate is predominantly used for the control of serum phosphorus levels (hyperphosphatemia) in adult patients with Chronic Kidney Disease (CKD) on dialysis, and for the treatment of iron deficiency anemia (IDA) in adults with non-dialysis dependent CKD, owing to its unique dual-action mechanism.
How is the market size of Ferric Citrate projected to grow?
The Ferric Citrate Market is forecasted to exhibit robust growth, driven by the increasing global incidence of CKD and heightened demand for effective dual-action phosphate binders, projected to grow at a Compound Annual Growth Rate (CAGR) of 7.8% between 2026 and 2033.
Which geographical region holds the largest market share for Ferric Citrate?
North America currently holds the largest market share, attributed to favorable reimbursement systems, high prevalence of End-Stage Renal Disease (ESRD), and strong market penetration by proprietary formulations. Asia Pacific, however, is anticipated to be the fastest-growing region.
What are the main drivers influencing the adoption of Ferric Citrate?
Key drivers include the global rise in CKD cases linked to diabetes and hypertension, clinical guidelines supporting simultaneous treatment of hyperphosphatemia and iron deficiency, and the demonstrated efficacy of ferric citrate in reducing the need for intravenous iron therapy.
Who are the major competitors in the Ferric Citrate market?
The branded market is largely dominated by companies such as Akebia Therapeutics (Keryx Biopharmaceuticals). However, competition also arises from manufacturers of non-iron phosphate binders (e.g., Sevelamer and Lanthanum) and API manufacturers serving potential generic drug entrants.
To check our Table of Contents, please mail us at: sales@marketresearchupdate.com
Research Methodology
The Market Research Update offers technology-driven solutions and its full integration in the research process to be skilled at every step. We use diverse assets to produce the best results for our clients. The success of a research project is completely reliant on the research process adopted by the company. Market Research Update assists its clients to recognize opportunities by examining the global market and offering economic insights. We are proud of our extensive coverage that encompasses the understanding of numerous major industry domains.
Market Research Update provide consistency in our research report, also we provide on the part of the analysis of forecast across a gamut of coverage geographies and coverage. The research teams carry out primary and secondary research to implement and design the data collection procedure. The research team then analyzes data about the latest trends and major issues in reference to each industry and country. This helps to determine the anticipated market-related procedures in the future. The company offers technology-driven solutions and its full incorporation in the research method to be skilled at each step.
The Company's Research Process Has the Following Advantages:
- Information Procurement
The step comprises the procurement of market-related information or data via different methodologies & sources.
- Information Investigation
This step comprises the mapping and investigation of all the information procured from the earlier step. It also includes the analysis of data differences observed across numerous data sources.
- Highly Authentic Source
We offer highly authentic information from numerous sources. To fulfills the client’s requirement.
- Market Formulation
This step entails the placement of data points at suitable market spaces in an effort to assume possible conclusions. Analyst viewpoint and subject matter specialist based examining the form of market sizing also plays an essential role in this step.
- Validation & Publishing of Information
Validation is a significant step in the procedure. Validation via an intricately designed procedure assists us to conclude data-points to be used for final calculations.
×
Request Free Sample:
Related Reports
- Ammonium Ferric Citrate Market Size, By Region (North America, Europe, Asia-Pacific, Latin America, Middle East and Africa), By Statistics, Trends, Outlook and Forecast 2026 to 2033
- Ferric Citrate Market Size Report By Type (Capsules, Tablets , Oral Solution), By Application (Medicine, Food & Nutritional Supplement, Others), By Region (North America, Latin America, Europe, Asia Pacific, Middle East, and Africa) - Share, Trends, Outlook and Forecast 2025-2032
Select License

Why Choose Us

We're cost-effective and Offered Best services:
We are flexible and responsive startup research firm. We adapt as your research requires change, with cost-effectiveness and highly researched report that larger companies can't match.

Information Safety
Market Research Update ensure that we deliver best reports. We care about the confidential and personal information quality, safety, of reports. We use Authorize secure payment process.

We Are Committed to Quality and Deadlines
We offer quality of reports within deadlines. We've worked hard to find the best ways to offer our customers results-oriented and process driven consulting services.

Our Remarkable Track Record
We concentrate on developing lasting and strong client relationship. At present, we hold numerous preferred relationships with industry leading firms that have relied on us constantly for their research requirements.

Best Service Assured
Buy reports from our executives that best suits your need and helps you stay ahead of the competition.

Customized Research Reports
Our research services are custom-made especially to you and your firm in order to discover practical growth recommendations and strategies. We don't stick to a one size fits all strategy. We appreciate that your business has particular research necessities.

Service Assurance
At Market Research Update, we are dedicated to offer the best probable recommendations and service to all our clients. You will be able to speak to experienced analyst who will be aware of your research requirements precisely.
Contact With Our Sales Team
Customer Testimonials
The content of the report is always up to the mark. Good to see speakers from expertise authorities.
Privacy requested , Managing Director
A lot of unique and interesting topics which are described in good manner.
Privacy requested, President
Well researched, expertise analysts, well organized, concrete and current topics delivered in time.
Privacy requested, Development Manager