Medical Disinfection and Sterilization Equipment Market Size, By Region (North America, Europe, Asia-Pacific, Latin America, Middle East and Africa), By Statistics, Trends, Outlook and Forecast 2026 to 2033 (Financial Impact Analysis)
ID : MRU_ 444644 | Date : Feb, 2026 | Pages : 258 | Region : Global | Publisher : MRU
Medical Disinfection and Sterilization Equipment Market Size
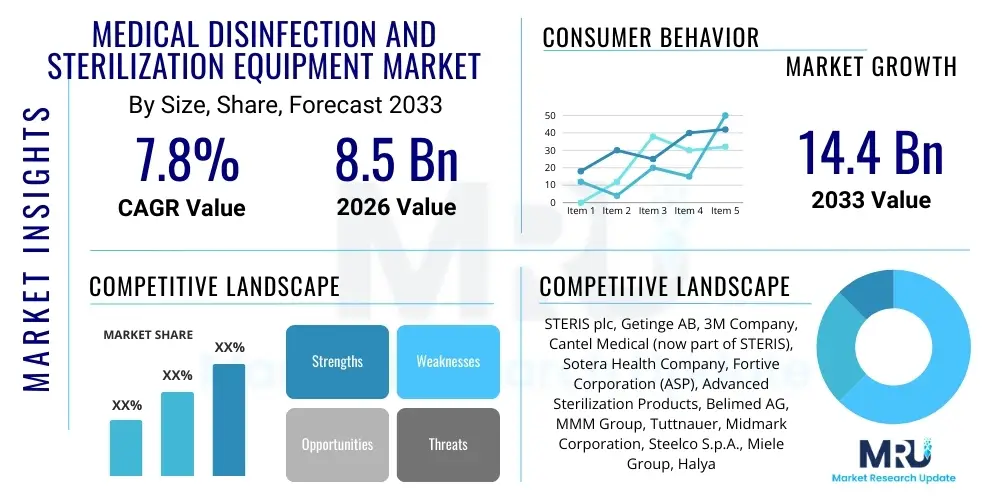
The Medical Disinfection and Sterilization Equipment Market is projected to grow at a Compound Annual Growth Rate (CAGR) of 7.8% between 2026 and 2033. The market is estimated at USD 13.5 Billion in 2026 and is projected to reach USD 23.2 Billion by the end of the forecast period in 2033. This robust growth trajectory is primarily fueled by the escalating prevalence of hospital-acquired infections (HAIs), the increasing volume of surgical procedures worldwide, and the stringent regulatory frameworks mandating high standards of cleanliness and asepsis in healthcare facilities. Advances in medical technology, including the proliferation of complex surgical instruments and devices, necessitate sophisticated disinfection and sterilization solutions, further driving market expansion. Furthermore, the global aging population and the associated rise in chronic diseases contribute to a greater demand for healthcare services, which in turn amplifies the need for effective infection control measures across various healthcare settings, from large hospitals to outpatient clinics and dental practices, cementing the market's significant upward trend.
Medical Disinfection and Sterilization Equipment Market introduction
The Medical Disinfection and Sterilization Equipment Market encompasses a broad spectrum of devices, instruments, and consumables crucial for preventing the spread of infections in healthcare environments. This market is fundamental to patient safety and public health, offering solutions that range from high-level disinfection for heat-sensitive instruments to complete sterilization for surgical tools and implantable devices. The primary objective is to eliminate or reduce microbial contamination on surfaces, medical devices, and in the air, thereby mitigating the risk of healthcare-associated infections (HAIs). Products within this market include autoclaves, ethylene oxide sterilizers, hydrogen peroxide gas plasma sterilizers, and various disinfectants such as glutaraldehyde, peracetic acid, and advanced enzymatic cleaners. The continuous innovation in these technologies aims to enhance efficacy, reduce processing times, and improve safety for both patients and healthcare personnel, addressing the evolving challenges in infection control.
Major applications of medical disinfection and sterilization equipment span across hospitals, clinics, diagnostic centers, research laboratories, and pharmaceutical companies. In hospitals, these systems are indispensable in operating theaters, intensive care units, and central sterile supply departments (CSSDs) to ensure all reusable medical instruments are free from pathogens before use. Beyond surgical instruments, the equipment is utilized for sterilizing laboratory apparatus, dental tools, endoscopes, and various other critical and semi-critical medical devices. The scope of application is expanding to include more complex and intricate devices, demanding innovative approaches to ensure thorough decontamination without compromising the integrity of sensitive materials. The continuous demand across these diverse settings underscores the universal importance of infection prevention in modern healthcare delivery systems.
The benefits derived from advanced disinfection and sterilization equipment are multifaceted and critical to healthcare system integrity. Foremost among these is the dramatic reduction in healthcare-associated infection rates, which not only saves lives but also lowers healthcare costs associated with extended hospital stays and treatment for infections. Effective sterilization processes enhance patient outcomes, build trust in medical procedures, and ensure compliance with stringent regulatory guidelines set by bodies such as the FDA, CDC, and WHO. Driving factors for this market's growth include the rising global incidence of infectious diseases, increasing awareness regarding infection control among healthcare professionals and the public, and technological advancements leading to more efficient, safer, and environmentally friendly sterilization methods. Furthermore, the expansion of healthcare infrastructure, particularly in emerging economies, coupled with increased investments in healthcare safety measures, continues to propel the market forward, ensuring robust demand for these essential medical technologies.
Medical Disinfection and Sterilization Equipment Market Executive Summary
The Medical Disinfection and Sterilization Equipment Market is experiencing substantial growth, propelled by a confluence of critical business trends. A prominent trend is the increasing adoption of automated and low-temperature sterilization methods, driven by the need to process heat-sensitive medical devices more efficiently and safely. Healthcare facilities are increasingly investing in advanced systems like hydrogen peroxide gas plasma and vaporized hydrogen peroxide sterilizers, which offer faster cycle times and reduced environmental impact compared to traditional methods. Furthermore, there is a growing emphasis on outsourcing sterilization services to specialized third-party providers, particularly among smaller clinics and ambulatory surgical centers, to manage costs and ensure compliance with complex regulatory standards. The development of integrated disinfection solutions that combine cleaning, disinfection, and sterilization in a streamlined workflow is also gaining traction, optimizing operational efficiency and reducing human error in infection control processes. These evolving business models and technological shifts are redefining market dynamics.
Regional trends significantly shape the market landscape. North America and Europe currently dominate the market, attributed to their highly developed healthcare infrastructures, stringent regulatory environments, and high adoption rates of advanced medical technologies. These regions are characterized by a strong presence of key market players and significant research and development investments aimed at pioneering novel disinfection and sterilization techniques. However, the Asia Pacific region is projected to exhibit the highest growth rate during the forecast period. This rapid expansion is fueled by improving healthcare access, increasing healthcare expenditure, a burgeoning medical tourism sector, and a rising awareness of infection control in developing economies such like China and India. Latin America, the Middle East, and Africa are also emerging as promising markets, driven by healthcare infrastructure development initiatives and a growing focus on preventing infectious diseases, though they still face challenges related to infrastructure and affordability. The global distribution of HAIs and evolving healthcare standards continue to dictate regional investment priorities and market development.
Segmentation trends reveal distinct patterns within the Medical Disinfection and Sterilization Equipment Market. By product type, sterilization equipment holds the largest share due to its indispensable role in rendering critical medical devices safe for use. Within this segment, steam sterilizers (autoclaves) remain foundational, while advanced low-temperature sterilizers are rapidly gaining ground. Disinfectants and sterilization accessories also represent significant and growing sub-segments, driven by continuous usage and replacement needs. By end-user, hospitals consistently account for the largest market share, given their extensive surgical volumes and high patient turnover requiring comprehensive infection control. However, the ambulatory surgical centers and clinics segment is expected to grow at an accelerated pace, reflecting the shift towards outpatient care and increasing procedural volumes in these settings. Furthermore, pharmaceutical and medical device companies contribute significantly as they require stringent sterilization for their products during manufacturing. These segment-specific dynamics underscore the diverse needs and operational models across the healthcare ecosystem, influencing product innovation and market penetration strategies.
AI Impact Analysis on Medical Disinfection and Sterilization Equipment Market
User inquiries regarding AI's influence on the Medical Disinfection and Sterilization Equipment Market frequently revolve around automation, predictive maintenance, enhanced efficacy, and improved compliance. Users are keen to understand how AI can reduce human error in complex sterilization processes, predict equipment malfunctions before they occur, and optimize workflow efficiency in central sterile supply departments (CSSDs). There is significant interest in AI's potential to analyze vast datasets from sterilization cycles to identify patterns, ensure optimal parameters, and even adapt protocols for new or challenging medical devices. Concerns often include the initial cost of integrating AI systems, the need for specialized training for healthcare staff, and ensuring data privacy and security within these advanced platforms. Expectations are high for AI to transform infection control from a reactive process into a proactive, intelligent system that consistently delivers superior patient safety outcomes and operational excellence, minimizing the burden of HAIs and improving overall healthcare quality. The consensus is that AI will drive a paradigm shift in how sterilization and disinfection practices are managed and executed, leading to more robust and reliable infection prevention strategies.
- Automated Workflow Optimization: AI algorithms can streamline sterilization processes in CSSDs, optimizing the loading of sterilizers, scheduling cycles, and managing inventory of sterile instruments, significantly reducing turnaround times and human intervention.
- Predictive Maintenance: AI-driven analytics can monitor equipment performance in real-time, predicting potential failures of sterilizers or disinfectors, thus enabling proactive maintenance and minimizing costly downtime while extending asset lifespan.
- Enhanced Efficacy and Validation: AI can analyze extensive data from sterilization cycles (temperature, pressure, exposure time) to ensure optimal parameters are met for different instrument types, enhancing the consistency and reliability of sterilization outcomes and improving validation processes.
- Smart Disinfectant Dispensing: AI-powered systems can precisely control the dispensing of disinfectants, ensuring correct concentrations and contact times for various surfaces and instruments, reducing chemical waste and improving effectiveness.
- Intelligent Inventory Management: AI can forecast demand for sterile instruments and disinfectants based on surgical schedules and patient flow, optimizing inventory levels, reducing stockouts, and preventing overstocking, leading to cost savings.
- Training and Compliance Monitoring: AI platforms can offer personalized training modules for healthcare staff on sterilization protocols and continuously monitor adherence to regulatory guidelines, flagging deviations and improving overall compliance with infection control standards.
- Robotics Integration: AI-powered robots can assist in handling and sorting contaminated instruments, reducing human exposure to hazardous materials and improving efficiency in the cleaning and preparation stages of sterilization.
DRO & Impact Forces Of Medical Disinfection and Sterilization Equipment Market
The Medical Disinfection and Sterilization Equipment Market is propelled by several robust drivers. A primary driver is the escalating prevalence of healthcare-associated infections (HAIs) globally, which places significant burden on healthcare systems and necessitates stringent infection control measures. The increasing volume of surgical procedures, including minimally invasive surgeries and complex implantable device operations, directly translates into a higher demand for sterile medical instruments. Furthermore, the aging global population and the concomitant rise in chronic diseases contribute to more frequent hospital visits and medical interventions, thereby intensifying the need for effective disinfection and sterilization. Stringent regulatory guidelines and recommendations from international bodies like the WHO, CDC, and local health authorities also mandate the adoption of advanced sterilization technologies, ensuring compliance and enhancing patient safety standards across diverse healthcare settings. Technological advancements leading to more efficient, safer, and faster sterilization methods further accelerate market growth by offering superior solutions to healthcare providers.
Conversely, the market faces significant restraints. The high initial capital investment required for purchasing advanced sterilization equipment, particularly for large-scale systems like automated central sterile supply department (CSSD) installations, can be prohibitive for smaller healthcare facilities or those in developing economies with limited budgets. Operational challenges such as the need for skilled personnel to operate and maintain sophisticated equipment, as well as the complexity of validating and monitoring sterilization processes, also impede market growth. Moreover, the increasing cost of consumables, including sterilization wrappers, chemical indicators, and biological indicators, adds to the operational burden. Concerns regarding the environmental impact of certain sterilization agents, such as ethylene oxide, lead to ongoing efforts to find safer alternatives, which can slow down adoption of established technologies or increase R&D costs. The challenges in ensuring full compliance with evolving regulatory standards across various geographic regions also pose a constant hurdle for manufacturers and healthcare providers alike.
Despite the restraints, significant opportunities abound within the Medical Disinfection and Sterilization Equipment Market. The ongoing shift towards low-temperature sterilization methods presents a substantial growth avenue, driven by the increasing use of heat-sensitive medical devices, particularly in endoscopy and robotic surgery. Emerging economies, with their rapidly expanding healthcare infrastructures and rising awareness of infection control, represent untapped markets with immense growth potential. The development of advanced, integrated sterilization solutions that offer automation, data connectivity, and remote monitoring capabilities can enhance efficiency and reduce human error, attracting significant investment. Furthermore, the growing trend of contract sterilization services provides an opportunity for specialized providers to cater to facilities that lack the resources or expertise for in-house sterilization. Innovation in environmentally friendly and safer sterilization chemicals and technologies also presents a lucrative pathway, aligning with global sustainability goals. The continuous threat of new infectious diseases and antibiotic resistance further underscores the long-term, indispensable need for robust disinfection and sterilization solutions, solidifying future growth prospects.
Segmentation Analysis
The Medical Disinfection and Sterilization Equipment Market is comprehensively segmented to provide a detailed understanding of its diverse components and dynamics. This segmentation helps in identifying key growth areas, understanding competitive landscapes, and tailoring strategic approaches for various product types, end-users, and technologies. The market is primarily categorized by Product (Disinfection Equipment, Sterilization Equipment, Disinfectants & Sterilization Accessories), by End-User (Hospitals & Clinics, Pharmaceutical Companies, Medical Device Companies, Diagnostic Centers & Laboratories), and by Type of Disinfectant/Sterilant. Each segment offers unique insights into demand patterns, technological preferences, and market penetration strategies, reflecting the varied needs of healthcare providers and medical product manufacturers across the globe. Analyzing these segments provides a granular view of the market’s current structure and future growth trajectories.
- By Product
- Sterilization Equipment
- Heat Sterilization Equipment (e.g., Autoclaves/Steam Sterilizers, Dry Heat Sterilizers)
- Low-Temperature Sterilization Equipment (e.g., Ethylene Oxide (EtO) Sterilizers, Hydrogen Peroxide Gas Plasma Sterilizers, Vaporized Hydrogen Peroxide (VHP) Sterilizers, Formaldehyde Sterilizers, Ozone Sterilizers)
- Radiation Sterilization Equipment (e.g., Gamma Sterilization, E-beam Sterilization)
- Disinfection Equipment
- Washing & Disinfection Equipment (e.g., Washer-Disinfectors, Ultrasonic Cleaners)
- Endoscope Reprocessors
- UV Disinfection Systems
- Air Purification & Disinfection Systems
- Surface Disinfection Machines
- Disinfectants & Sterilization Accessories
- High-Level Disinfectants (e.g., Glutaraldehyde, Ortho-phthalaldehyde (OPA), Hydrogen Peroxide, Peracetic Acid)
- Intermediate & Low-Level Disinfectants (e.g., Quaternary Ammonium Compounds, Alcohols, Phenolics, Chlorine Compounds)
- Sterilization Accessories (e.g., Sterilization Indicators, Chemical Indicators, Biological Indicators, Sterilization Pouches & Wraps, Sterilization Containers, Filtration Systems)
- Enzymatic Cleaners
- Sterilization Equipment
- By End-User
- Hospitals & Clinics (including ASCs - Ambulatory Surgical Centers)
- Pharmaceutical Companies
- Medical Device Companies
- Diagnostic Centers & Laboratories
- Research & Academic Institutes
- Contract Research Organizations (CROs)
- Dental Clinics
- Food & Beverage Industry (relevant for certain sterilization applications)
- By Type of Disinfectant/Sterilant
- Chemical Disinfectants (e.g., Aldehydes, Alcohols, Phenols, Oxidizing Agents)
- Heat/Steam
- Low-Temperature Gases/Vapors (e.g., EtO, H2O2)
- Radiation
Value Chain Analysis For Medical Disinfection and Sterilization Equipment Market
The value chain for the Medical Disinfection and Sterilization Equipment Market is a complex ecosystem, beginning with upstream raw material suppliers and extending through manufacturing, distribution, and end-user deployment. Upstream analysis involves the procurement of essential components and chemicals. This includes specialized metals for sterilization equipment manufacturing, polymers for various consumables, and raw chemical compounds for disinfectant formulations. Key suppliers in this stage provide advanced electronics for control systems, durable materials for chamber construction, and precision engineering components for moving parts. The quality and cost-effectiveness of these raw materials directly impact the final product's performance, durability, and market competitiveness. Manufacturers often forge long-term relationships with reliable suppliers to ensure a consistent supply of high-quality inputs, as any disruption can have significant repercussions on production schedules and product availability within the critical healthcare sector.
The manufacturing stage is central to the value chain, where raw materials are transformed into finished medical disinfection and sterilization equipment and consumables. This involves design, engineering, assembly, and rigorous testing processes to ensure compliance with international quality and safety standards such as ISO 13485 and FDA regulations. Manufacturers invest heavily in research and development to innovate new technologies, improve efficiency, and reduce the environmental impact of their products. This stage also includes the formulation and production of various disinfectants and sterilization accessories. Key players in this segment differentiate themselves through technological superiority, product reliability, and comprehensive after-sales support. The complexity of these devices necessitates specialized manufacturing processes and a highly skilled workforce, emphasizing the importance of intellectual property and process optimization for competitive advantage in a demanding market.
Downstream analysis focuses on the distribution channels and the ultimate end-users of these critical products. Distribution primarily occurs through a mix of direct sales forces, third-party distributors, and increasingly, e-commerce platforms for consumables and smaller devices. Direct sales are often preferred for large capital equipment, allowing for specialized consultations, installation, and training services. Distributors play a crucial role in reaching a broader customer base, particularly in geographically diverse and emerging markets, offering logistics, local support, and streamlined procurement. End-users, primarily hospitals, ambulatory surgical centers, clinics, and pharmaceutical companies, represent the final consumption point. The effectiveness of the distribution channel is critical for ensuring timely delivery, product accessibility, and responsive technical support. The entire value chain is driven by the imperative of patient safety and infection prevention, with continuous feedback from end-users influencing product development and service improvements to meet evolving healthcare needs and regulatory requirements.
Medical Disinfection and Sterilization Equipment Market Potential Customers
The Medical Disinfection and Sterilization Equipment Market serves a diverse and expansive customer base, each segment having unique requirements for infection control and varying scales of operation. Hospitals, particularly large multi-specialty and university-affiliated institutions, represent the largest segment of potential customers. These facilities perform a high volume of complex surgical procedures, house numerous intensive care units, and manage a vast array of reusable medical devices, necessitating comprehensive and robust sterilization infrastructure. Central Sterile Supply Departments (CSSDs) within hospitals are critical hubs for processing and preparing instruments for all medical and surgical departments. Their demand spans across high-capacity autoclaves, advanced low-temperature sterilizers for heat-sensitive instruments, washer-disinfectors, and a continuous supply of sterilization consumables such as indicators, wraps, and disinfectants. The imperative to prevent healthcare-associated infections (HAIs) and comply with stringent regulatory standards drives their persistent demand for advanced and reliable equipment and services.
Ambulatory surgical centers (ASCs) and outpatient clinics constitute a rapidly growing segment of potential customers. As healthcare shifts towards more cost-effective and convenient outpatient settings, the volume of procedures performed in ASCs is steadily increasing. While typically smaller than hospitals, these facilities still require efficient and compliant sterilization solutions for their surgical instruments and other medical devices. Their needs often lean towards compact, fast-cycle sterilization equipment, and efficient disinfection systems for endoscopes and other semi-critical devices. Dental clinics also fall into this category, requiring specific sterilizers for dental handpieces and instruments, alongside surface disinfectants. These smaller establishments often seek user-friendly equipment with lower operational costs and compact footprints, yet without compromising on sterilization efficacy and regulatory adherence. The expansion of these settings globally underscores a significant and evolving customer base with distinct purchasing criteria.
Beyond direct patient care facilities, the market also serves other crucial entities within the healthcare and life sciences ecosystem. Pharmaceutical and biotechnology companies are significant customers, as they require sterile environments and equipment for drug manufacturing, research, and development. This includes sterilization of media, components, and equipment used in sterile compounding and biopharmaceutical production. Similarly, medical device manufacturers utilize advanced sterilization techniques for their products before packaging and distribution, ensuring sterility and patient safety. Diagnostic laboratories, research institutions, and academic centers also demand specialized disinfection and sterilization equipment for processing samples, culturing microorganisms, and maintaining sterile conditions for experiments. Furthermore, emerging markets in veterinary clinics and certain industrial applications (e.g., food and beverage processing, cosmetics manufacturing) that require aseptic conditions also represent potential, albeit niche, customer segments for specific types of disinfection and sterilization technologies and consumables, broadening the overall market reach.
| Report Attributes | Report Details |
|---|---|
| Market Size in 2026 | USD 13.5 Billion |
| Market Forecast in 2033 | USD 23.2 Billion |
| Growth Rate | 7.8% CAGR |
| Historical Year | 2019 to 2024 |
| Base Year | 2025 |
| Forecast Year | 2026 - 2033 |
| DRO & Impact Forces |
|
| Segments Covered |
|
| Key Companies Covered | 3M Company, Advanced Sterilization Products (ASP), Cantel Medical (a STERIS company), ECOLAB, Getinge AB, Matachana Group, MELAG Medizintechnik GmbH & Co. KG, Miele Professional, Nanosonics Ltd., Oro Clean Chemie AG, PENTAX Medical (a HOYA Company), Sakura Finetek Japan Co., Ltd., SCICAN Inc., Sotera Health (Sterigenics, Nordion, Nelson Labs), STERIS PLC, Steelco S.p.A., Terragene, Tuttnauer, TSO3 (a STERIS company), Zimmer Biomet (Biomet's sterilization products) |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Enquiry Before Buy | Have specific requirements? Send us your enquiry before purchase to get customized research options. Request For Enquiry Before Buy |
Medical Disinfection and Sterilization Equipment Market Key Technology Landscape
The technological landscape of the Medical Disinfection and Sterilization Equipment Market is characterized by continuous innovation aimed at enhancing efficacy, safety, speed, and material compatibility. One of the most significant advancements is the proliferation of low-temperature sterilization technologies, which are crucial for processing heat-sensitive instruments like endoscopes, complex surgical tools, and delicate electronic devices without causing damage. Key technologies in this segment include hydrogen peroxide gas plasma sterilization, vaporized hydrogen peroxide (VHP) systems, and ethylene oxide (EtO) sterilization. These methods offer distinct advantages in terms of broad-spectrum microbial kill, relatively short cycle times, and reduced material degradation compared to traditional heat-based methods. Manufacturers are also focusing on developing integrated systems that combine cleaning, disinfection, and sterilization into automated, validated workflows, minimizing human intervention and ensuring consistent results, a critical aspect in modern infection prevention protocols.
Beyond low-temperature methods, advancements in traditional steam sterilization (autoclaves) continue to optimize their performance. Modern autoclaves feature sophisticated control systems, faster pre-vacuum and post-vacuum cycles, and enhanced drying capabilities, making them more energy-efficient and reducing overall processing times. Washer-disinfectors, essential for the initial cleaning and high-level disinfection of reusable instruments, are evolving with improved enzymatic cleaning agents, ultrasonic technology, and customizable washing programs to tackle bioburden effectively. Furthermore, ultraviolet (UV-C) disinfection systems are gaining prominence for surface and air disinfection, especially in hospital rooms and operating theaters, offering a chemical-free and rapid method to reduce microbial load. These technologies are often integrated with smart features and connectivity options, allowing for remote monitoring, data logging, and seamless integration into hospital information systems for improved traceability and compliance.
The rapid integration of digital technologies, particularly IoT (Internet of Things) and AI (Artificial Intelligence), is fundamentally transforming the market. IoT sensors embedded in sterilization equipment enable real-time monitoring of critical parameters such as temperature, pressure, and chemical concentrations, ensuring optimal performance and compliance. AI-powered analytics can process this vast amount of data to predict equipment maintenance needs, optimize sterilization cycles based on instrument types, and identify potential issues before they escalate. This predictive capability enhances operational efficiency, reduces downtime, and ensures a higher level of safety and reliability. Furthermore, software solutions for instrument tracking and management are becoming increasingly sophisticated, providing end-to-end visibility of instruments through their entire reprocessing cycle. These technological convergences are not only improving the effectiveness of disinfection and sterilization processes but also contributing to greater cost-efficiency, regulatory adherence, and ultimately, enhanced patient safety outcomes across the entire healthcare spectrum.
Regional Highlights
- North America: The largest market share, driven by stringent regulatory frameworks (FDA, CDC), high healthcare expenditure, significant R&D investments, and the presence of major market players. The region exhibits high adoption rates of advanced low-temperature sterilization technologies and sophisticated automated systems in well-established healthcare infrastructures. The U.S. and Canada are at the forefront of implementing new infection control guidelines and leveraging technological innovations for enhanced patient safety, leading to sustained market dominance.
- Europe: The second-largest market, characterized by advanced healthcare systems, a strong emphasis on infection control standards (e.g., European Medical Device Regulation - MDR), and a growing aging population requiring frequent medical interventions. Countries like Germany, the UK, France, and Italy are key contributors, focusing on sustainable and environmentally friendly sterilization solutions, and investing in advanced washer-disinfectors and endoscope reprocessing units. The presence of numerous global and regional manufacturers further solidifies its market position.
- Asia Pacific (APAC): Expected to be the fastest-growing market due to rapid improvements in healthcare infrastructure, increasing healthcare expenditure, a large population base, and rising awareness of hospital-acquired infections. Countries like China, India, Japan, and South Korea are witnessing significant investments in new hospitals and clinics, driving demand for both basic and advanced disinfection and sterilization equipment. The expansion of medical tourism and a growing middle class capable of affording better healthcare services are also key growth accelerators in this dynamic region.
- Latin America: An emerging market experiencing steady growth, fueled by government initiatives to improve healthcare access and quality, particularly in Brazil, Mexico, and Argentina. While still facing challenges in terms of infrastructure and affordability, the region is showing increasing adoption of modern sterilization techniques, driven by the need to combat infectious diseases and comply with evolving international healthcare standards. Investments in public and private healthcare facilities are gradually expanding the market for disinfection and sterilization products.
- Middle East and Africa (MEA): This region presents significant growth potential, particularly in the GCC countries (Saudi Arabia, UAE) due to high healthcare spending, medical tourism, and the development of world-class medical facilities. In Africa, while disparities exist, there is a growing focus on improving public health infrastructure and combating infectious diseases, which drives the demand for essential disinfection and sterilization equipment. Increasing awareness and investment in modern healthcare practices are expected to foster continued market expansion, albeit at varying paces across the diverse sub-regions.
Top Key Players
The market research report includes a detailed profile of leading stakeholders in the Medical Disinfection and Sterilization Equipment Market.- 3M Company
- Advanced Sterilization Products (ASP)
- Cantel Medical (a STERIS company)
- ECOLAB
- Getinge AB
- Matachana Group
- MELAG Medizintechnik GmbH & Co. KG
- Miele Professional
- Nanosonics Ltd.
- Oro Clean Chemie AG
- PENTAX Medical (a HOYA Company)
- Sakura Finetek Japan Co., Ltd.
- SCICAN Inc.
- Sotera Health (Sterigenics, Nordion, Nelson Labs)
- STERIS PLC
- Steelco S.p.A.
- Terragene
- Tuttnauer
- TSO3 (a STERIS company)
- Zimmer Biomet (Biomet's sterilization products)
Frequently Asked Questions
What is the primary driver for the growth of the Medical Disinfection and Sterilization Equipment Market?
The primary driver is the increasing global prevalence of healthcare-associated infections (HAIs), which necessitates stringent infection control measures and robust disinfection and sterilization protocols in all healthcare settings to ensure patient safety and reduce healthcare costs.
How is AI impacting the Medical Disinfection and Sterilization Equipment Market?
AI is significantly impacting the market by enabling advanced automation, predictive maintenance for equipment, enhanced efficacy validation through data analytics, and optimized workflow management in central sterile supply departments (CSSDs), leading to improved efficiency and reliability.
Which product segment holds the largest share in the Medical Disinfection and Sterilization Equipment Market?
Sterilization equipment, particularly heat and low-temperature sterilizers, holds the largest market share due to its indispensable role in rendering critical medical devices and surgical instruments safe for reuse, directly impacting patient safety in numerous procedures.
What are the key challenges faced by the Medical Disinfection and Sterilization Equipment Market?
Key challenges include the high initial capital investment for advanced equipment, the need for skilled personnel for operation and maintenance, the rising cost of sterilization consumables, and ongoing concerns about the environmental impact of certain sterilization agents, leading to a search for greener alternatives.
Which geographical region is expected to show the fastest growth in this market?
The Asia Pacific region is projected to exhibit the fastest growth, driven by rapidly improving healthcare infrastructure, increasing healthcare expenditure, a large and aging population, and heightened awareness regarding infection control in countries like China, India, and South Korea.
To check our Table of Contents, please mail us at: sales@marketresearchupdate.com
Research Methodology
The Market Research Update offers technology-driven solutions and its full integration in the research process to be skilled at every step. We use diverse assets to produce the best results for our clients. The success of a research project is completely reliant on the research process adopted by the company. Market Research Update assists its clients to recognize opportunities by examining the global market and offering economic insights. We are proud of our extensive coverage that encompasses the understanding of numerous major industry domains.
Market Research Update provide consistency in our research report, also we provide on the part of the analysis of forecast across a gamut of coverage geographies and coverage. The research teams carry out primary and secondary research to implement and design the data collection procedure. The research team then analyzes data about the latest trends and major issues in reference to each industry and country. This helps to determine the anticipated market-related procedures in the future. The company offers technology-driven solutions and its full incorporation in the research method to be skilled at each step.
The Company's Research Process Has the Following Advantages:
- Information Procurement
The step comprises the procurement of market-related information or data via different methodologies & sources.
- Information Investigation
This step comprises the mapping and investigation of all the information procured from the earlier step. It also includes the analysis of data differences observed across numerous data sources.
- Highly Authentic Source
We offer highly authentic information from numerous sources. To fulfills the client’s requirement.
- Market Formulation
This step entails the placement of data points at suitable market spaces in an effort to assume possible conclusions. Analyst viewpoint and subject matter specialist based examining the form of market sizing also plays an essential role in this step.
- Validation & Publishing of Information
Validation is a significant step in the procedure. Validation via an intricately designed procedure assists us to conclude data-points to be used for final calculations.
×
Request Free Sample:
Related Reports
Select License

Why Choose Us

We're cost-effective and Offered Best services:
We are flexible and responsive startup research firm. We adapt as your research requires change, with cost-effectiveness and highly researched report that larger companies can't match.

Information Safety
Market Research Update ensure that we deliver best reports. We care about the confidential and personal information quality, safety, of reports. We use Authorize secure payment process.

We Are Committed to Quality and Deadlines
We offer quality of reports within deadlines. We've worked hard to find the best ways to offer our customers results-oriented and process driven consulting services.

Our Remarkable Track Record
We concentrate on developing lasting and strong client relationship. At present, we hold numerous preferred relationships with industry leading firms that have relied on us constantly for their research requirements.

Best Service Assured
Buy reports from our executives that best suits your need and helps you stay ahead of the competition.

Customized Research Reports
Our research services are custom-made especially to you and your firm in order to discover practical growth recommendations and strategies. We don't stick to a one size fits all strategy. We appreciate that your business has particular research necessities.

Service Assurance
At Market Research Update, we are dedicated to offer the best probable recommendations and service to all our clients. You will be able to speak to experienced analyst who will be aware of your research requirements precisely.
Contact With Our Sales Team
Customer Testimonials
The content of the report is always up to the mark. Good to see speakers from expertise authorities.
Privacy requested , Managing Director
A lot of unique and interesting topics which are described in good manner.
Privacy requested, President
Well researched, expertise analysts, well organized, concrete and current topics delivered in time.
Privacy requested, Development Manager
